Advertisements
Advertisements
प्रश्न
Account for the following:
CH3CHO is more reactive than CH3COCH3 towards reaction with HCN.
उत्तर
CH3COCH3 is sterically hindered than CH3CHO due to the presence of alkyl group on both sides of the carbonyl carbon, making them less reactive towards nucleophilic attack because both methyl groups have electron releasing tendency due to -I effect. These alkyl groups make ketone less reactive by donating electron to carbonyl group.Therefore, acetaldehyde is more reactive towards reaction with HCN.
APPEARS IN
संबंधित प्रश्न
Give reasons for the following : Boiling point of ethanol is higher in comparison to methoxymethane.
Alcohols are comparatively more soluble in water than hydrocarbons of comparable molecular masses. Explain this fact.
Give simple chemical tests to distinguish between the following pairs of compounds :
Ethanol and phenol
Identify the chiral molecule in the following pair :
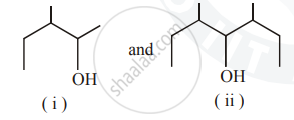
Alcohols have high boiling points because of ____________.
Which one of the following alcohols is least soluble in water?
Arrange the following compounds in increasing order of boiling point. Propan-1-ol, butan-1-ol, butan-2-ol, pentan-1-ol
Explain why alcohols and ethers of comparable molecular mass have different boiling points?
How are the following conversions carried out?
\[\ce{Methyl magnesium bromide -> 2-Methylpropan-2-ol}\]
How is the following conversion carried out?
\[\ce{Methyl magnesium bromide-> 2-Methylpropan-2-ol}\]
