Advertisements
Advertisements
प्रश्न
Answer the following.
Using Raoult’s law, how will you show that ΔP = `"P"_1^0 "x"_2`? Where, x2 is the mole fraction of solute in the solution and `"P"_1^0` vapour pressure of pure solvent.
उत्तर
1. Raoult’s law expresses the quantitative relationship between vapour pressure of solution and vapour pressure of the solvent.
2. In solutions of nonvolatile solutes, the law is applicable only to the volatile solvent.
3. The law states that, “the vapour pressure of solvent over the solution is equal to the vapour pressure of pure solvent multiplied by its mole fraction in the solution.”
4. Suppose that for a binary solution containing solvent and one nonvolatile solute, P1 is the vapour pressure of solvent over the solution, x1 and x2 are the mole fractions of solvent and solute, respectively and `"P"_1^0` is the vapour pressure of pure solvent, then, `"P"_1 = "P"_1^0 "x"_1`.
5. Since, x1 = 1 – x2,
`"P"_1 = "P"_1^0 "x"_1 = "P"_1^0 (1 - "x"_2) = "P"_1^0 - "P"_1^0 "x"_2`
∴ `"P"_1^0 - "P"_1 = "P"_1^0 "x"_2`
∴ `triangle "P" = "P"_1^0 "x"_2` (∵ Δ P is the lowering of vapour pressure)
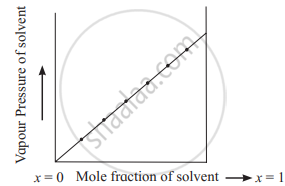
Note: A plot of P1 versus x1 is a straight as shown below.
Variation of vapour pressure of solution with mole fraction of solvent
APPEARS IN
संबंधित प्रश्न
Choose the most correct option.
The vapour pressure of a solution containing 2 moles of a solute in 2 moles of water (vapour pressure of pure water = 24 mm Hg) is ______.
Choose the most correct option.
Vapour pressure of a solution is _______.
Answer the following in one or two sentences.
State Raoult’s law for a solution containing a nonvolatile solute.
Fish generally needs O2 concentration in water at least 3.8 mg/L for survival. What partial pressure of O2 above the water is needed for the survival of fish? Given the solubility of O2 in water at 0 °C and 1 atm-1 partial pressure is 2.2 × 10-3 atm mol/L.
The vapour pressure of water at 20°C is 17 mm Hg. What is the vapour pressure of solution containing 2.8 g urea in 50 g of water?
A mixture of benzene and toluene contains 30% by mass of toluene. At 30 °C, vapour pressure of pure toluene is 36.7 mm Hg and that of pure benzene is 118.2 mm Hg. Assuming that the two liquids form ideal solutions, calculate the total pressure and partial pressure of each constituent above the solution at 30 °C.
State Raoult’s law.
What is enthalpy change and volume change of mixing of two components forming an ideal solution?
The vapour pressures of pure liquids A and B are 0.600 bar and 0.933 bar respectively, at a certain temperature.
What is the mole fraction of liquid B in the solution when the total vapour pressure of their mixture is 0.8 bar?
Distinguish between ideal and non-ideal solutions.
The vapour pressure of a pure solvent at a certain temperature is 0.0227 bar. What is the vapour pressure of a solution containing 6 g of solute (M = 60 g/mol) in 50 g of solvent?
What are nonideal solutions?
Explain with reasons and diagrams, the positive and negative deviations from Raoult’s law shown by nonideal solutions.
Which of the following statements is INCORRECT?
Which of the following metals is refined by vapour phase refining in Mond process?
If 1 mole of ____________ is added to 1 dm3 of water, the boiling point of water will decrease.
A solution of acetone in ethanol ______.
18 g of glucose (C6H12O6) is added to 178.2 g of water. The vapour pressure of water for this aqueous solution is ______.
For an ideal binary liquid solution with `"p"_"A"^circ > "P"_"B"^circ`, which is a relation between XA (mole fraction of A in liquid phase) and YA (mole fraction of A in vapour phase) is correct, XB and YB are mole fractions of B in liquid and vapour phase respectively?
What are ideal and non-ideal solutions?
Explain the variation of vapour pressures with mole fractions of binary solutions of two volatile liquids forming ideal solutions.
Write the relationship between mole fraction of solvent and vapour pressure of solution.
The solutions that obey Raoult's law over entire range of concentrations are ______.
