Advertisements
Advertisements
प्रश्न
Arrange the following in the increasing order of their reactivity towards nucleophilic addition reaction:
C6H5COCH3, CH3-CHO, CH3COCH3
उत्तर
Aldehydes are generally more reactive than ketones in nucleophilic addition reactions because of steric and electronic reasons. In case of ketones, aromatic ketones are more susceptible to nucleophilic attack than aliphatic ketones.
CH3COCH3 < C6H5COCH3 < CH3CHO
APPEARS IN
संबंधित प्रश्न
Predict the products of the following reactions:
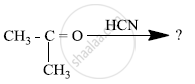
Arrange the following compound in increasing order of its reactivity in nucleophilic addition reactions.
Ethanal, Propanal, Propanone, Butanone.
Hint: Consider steric effect and electronic effect.
Acetaldehyde, Acetone, Di-tert-butyl ketone, Methyl tert-butyl ketone (reactivity towards HCN)
Give plausible explanation for the following:
There are two −NH2 groups in semicarbazide. However, only one is involved in the formation of semicarbazones.
Write balanced chemical equations for action of ammonia on - acetone
What happens when ethanal is treated with excess ethanol and acid?
Draw structures of the given derivatives.
The ethylene ketal of hexan-3-one
Draw the structure of the following derivative.
The ethylene ketal of hexane-3-one
Draw structure of the following derivative.
The ethylene ketal of hexane-3-one
Draw structures of the following derivative.
The ethylene ketal of hexan-3-one
