Advertisements
Advertisements
प्रश्न
Arrange the following as per the instruction given in the bracket:
Al, K, Mg, Ca (decreasing order of its reactivity)
उत्तर
K > Ca > Mg > Al
APPEARS IN
संबंधित प्रश्न
Write the chemical equation for the event.
A reaction was brought about between ferric oxide and aluminum.
Divide the metals Cu, Zn, Ca, Mg, Fe, Na, Li into three groups, namely reactive metals, moderately reactive metals and less reactive metals.
State what is meant by the ‘reactivity series of metals’
Select the correct answer for the statement given below:
The catalyst used in the catalytic reaction involving the reactants nitrogen and hydrogen.
Select the correct answer for the statement given below:
A neutral oxide which does not react with an acid or a base to give salt and water.
Explain the following reaction with the balanced equation.
Sodium burns in air
Observe the following diagram and identify the type of reaction and write observation.
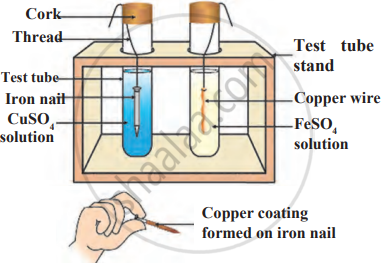
A metal that exists as a liquid at room temperature is obtained by heating its sulphide in the presence of air. Identify the metal and its ore and give the reaction involved.
Of the three metals X, Y and Z. X reacts with cold water, Y with hot water and Z with steam only. Identify X, Y and Z and also arrange them in order of increasing reactivity.
An element A burns with golden flame in air. It reacts with another element B, atomic number 17 to give a product C. An aqueous solution of product C on electrolysis gives a compound D and liberates hydrogen. Identify A, B, C and D. Also write down the equations for the reactions involved.
