Advertisements
Advertisements
Question
Arrange the following as per the instruction given in the bracket:
Al, K, Mg, Ca (decreasing order of its reactivity)
Solution
K > Ca > Mg > Al
APPEARS IN
RELATED QUESTIONS
How will you obtain Magnesium oxide from magnesium.
Also give balanced equations for the reactions
Write chemical equation for the event.
Aluminium came in contact with air.
Write the chemical equation for the event.
A reaction was brought about between ferric oxide and aluminum.
With reference to Water explain with suitable examples of how the reactivity of the metals could be differentiated.
Complete the statement by filling in the blank with the correct word:
The metal which reacts with steam and the reaction is reversible is ________.
Explain the following reaction with the balanced equation.
Reaction of aluminium with oxygen
Explain the following reaction with the balanced equation.
Sulphur burns in air
Observe the following diagram and identify the type of reaction and write observation.
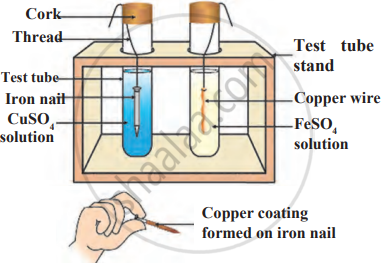
A metal that exists as a liquid at room temperature is obtained by heating its sulphide in the presence of air. Identify the metal and its ore and give the reaction involved.
Of the three metals X, Y and Z. X reacts with cold water, Y with hot water and Z with steam only. Identify X, Y and Z and also arrange them in order of increasing reactivity.
