Advertisements
Advertisements
Question
With reference to Water explain with suitable examples of how the reactivity of the metals could be differentiated.
Solution
Action of metals with water: K, Na, Ca react with cold water.
K: Darts on the water surface and react violently.
Na: Revolves on the water surface and bums.
Ca: Sinks in water, react less violently.
Hence, K > Na > Ca calcium is less reactive than K and Na.
Mg and Al react with boiling water/steam.
Hence, Mg and Al are less reactive than K, Na, and Ca. Zn, Fe, Pb also react with steam and reaction stops soon. Fe when hot reacts with steam
This shows that Mg > Al > Zn > Fe > Pb.
Cu, Hg, Ag, Pt, Au do not react with steam or even when hot is less reactive.
APPEARS IN
RELATED QUESTIONS
Fill in the blank
When a piece of copper is added to silver nitrate solution, it turns ............in colour.
What is meant by the metal reactivity series ? State its importance, (any two points).
Write chemical equation for the event.
Iron filings are dropped in aqueous solution of copper sulphate.
Select the correct answer for the statement given below:
A neutral oxide which does not react with an acid or a base to give salt and water.
In preparation of Aqua regia hydrochloric acid and _______ acid are mixed.
Classify the following metals based on their reactivity.
Cu, Zn, Ca, Mg, Fe, Na, Li, Hg
| More reactive | Moderately reactive | Less reactive |
Explain the following reaction with the balanced equation.
Magnesium reacts with dil HCl
Observe the following diagram and identify the type of reaction and write observation.
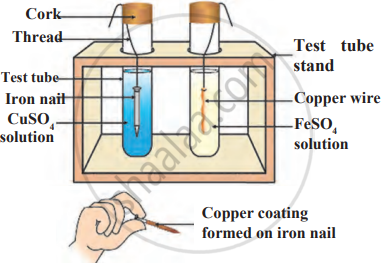
Which among the following alloys contain mercury as one of its constituents?
Compound X and aluminium are used to join railway tracks.
- Identify the compound X
- Name the reaction
- Write down its reaction.
