Advertisements
Advertisements
Question
Select the correct answer for the statement given below:
A neutral oxide which does not react with an acid or a base to give salt and water.
Options
Iron
Carbonic acid
Hydrogen
Oxygen
Carbon monoxide
Solution
Carbon monoxide
APPEARS IN
RELATED QUESTIONS
Write chemical equation for the event.
Aluminium came in contact with air.
Write chemical equation for the event.
Electrolysis of alumina is done.
Give a balanced equation for the following type of reaction:
A displacement reaction in which a metal above hydrogen in the reactivity series, displaces another metal from the solution of its compound.
Classify the following metals based on their reactivity.
Cu, Zn, Ca, Mg, Fe, Na, Li, Hg
| More reactive | Moderately reactive | Less reactive |
Explain the following reaction with the balanced equation.
Magnesium reacts with dil HCl
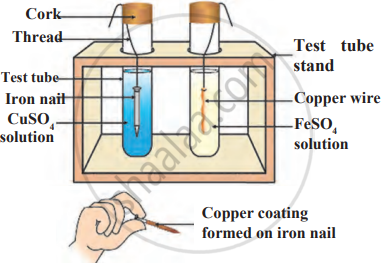
Observe the following diagram and identify the type of reaction and write observation.
Which among the following statements is incorrect for magnesium metal?
Which among the following alloys contain mercury as one of its constituents?
A metal M does not liberate hydrogen from acids but reacts with oxygen to give a black colour product. Identify M and black coloured product and also explain the reaction of M with oxygen.
Arrange the following as per the instruction given in the bracket:
Al, K, Mg, Ca (decreasing order of its reactivity)
