Advertisements
Advertisements
Question
Compound X and aluminium are used to join railway tracks.
- Identify the compound X
- Name the reaction
- Write down its reaction.
Solution
- Compound X is Fe2O3
- Displacement reaction
- `"Fe"_2"O"_3("s") + 2"Al"overset("Heat")(->)"Al"_2"O"_3("s") + 2"Fe"("l")`
APPEARS IN
RELATED QUESTIONS
What do you observe when silver nitrate is added to a solution of sodium chloride?
What do you observe when ferrous sulphate solution is added to an aqueous solution of sodium hydroxide.
How will you obtain Magnesium oxide from magnesium.
Also give balanced equations for the reactions
How will you obtain Silver chloride from silver nitrate.
Also give balanced equations for the reactions
Give a balanced equation for the following type of reaction:
A displacement reaction in which a metal above hydrogen in the reactivity series, displaces another metal from the solution of its compound.
Explain the following reaction with the balanced equation.
Sodium burns in air
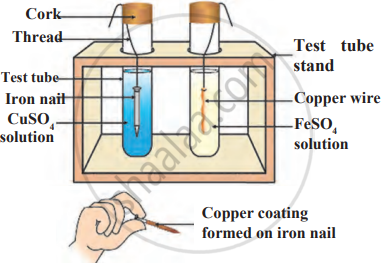
Observe the following diagram and identify the type of reaction and write observation.
A metal A, which is used in thermite process, when heated with oxygen gives an oxide B, which is amphoteric in nature. Identify A and B. Write down the reactions of oxide B with HCl and NaOH.
A metal that exists as a liquid at room temperature is obtained by heating its sulphide in the presence of air. Identify the metal and its ore and give the reaction involved.
A metal M does not liberate hydrogen from acids but reacts with oxygen to give a black colour product. Identify M and black coloured product and also explain the reaction of M with oxygen.
