Advertisements
Advertisements
Question
When a metal X is treated with cold water, it gives a basic salt Y with molecular formula XOH (Molecular mass = 40) and liberates a gas Z which easily catches fire. Identify X, Y and Z and also write the reaction involved.
Solution
When sodium metal is treated with cold water, it gives a base sodium hydroxide and hydrogen gas is liberated which undergo explosive combustion reaction in an atmosphere of oxygen gas with a pop sound.
Hence, metal X is sodium, base Y is sodium hydroxide and gas Z is hydrogen gas.
`2"Na"("s") + 2"H"_2"O"("l") -> 2"NaOH"("aq") + "H"_2("g")`
APPEARS IN
RELATED QUESTIONS
Give two examples of amphoteric oxides.
Which of the following oxide(s) of iron would be obtained on the prolonged reaction of iron with steam?
Which of the following oxide(s) of iron would be obtained on prolonged reaction of iron with steam?
Stainless steel is very useful material for our life. In stainless steel, iron is mixed with
If copper is kept open in air, it slowly loses its shining brown surface and gains a green coating. It is due to the formation of
Metals generally react with dilute acids to produce hydrogen gas. Which one of the following metals does not react with dilute hydrochloric acid?
Which of the following reacts with cold water vigorously?
Answer the questions based on the figure below:
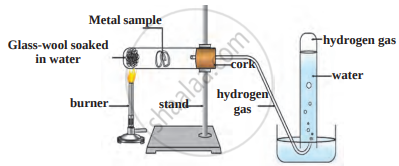
- Which reaction is shown in the figure?
- Which gas is evolved in the reaction?
- Give an example of reactants that rapidly show this reaction. Give equations.
- Give an example of reactants that do not react rapidly.
- In what condition will reactants of (c) part react? Give equation.
Select the appropriate state symbols of the products given as X and Y in the following chemical equation by choosing the correct option from table given below:
\[\ce{Zn_{(s)} + H_2SO_{4(l)} ->ZnSO_{4(X)} + H_{2(Y)}}\]
Write a chemical equation showing the ionic products formed on dissolving potassium hydroxide in water.
