Advertisements
Advertisements
Question
Metals generally react with dilute acids to produce hydrogen gas. Which one of the following metals does not react with dilute hydrochloric acid?
Options
Magnesium
Aluminium
Iron
Copper
Solution
Copper
Explanation -
The less reactive metals like copper, silver and gold do not react with dilute hydrochloric acid to produce hydrogen gas.
APPEARS IN
RELATED QUESTIONS
Give two examples of amphoteric oxides.
What are amphoteric oxides?
Which of the following metals liberate hydrogen with 5% HNO3?
(i) Cu
(ii) Zn
(iii) Mn
(iv) Mg
Which of the following oxide(s) of iron would be obtained on prolonged reaction of iron with steam?
When a metal X is treated with cold water, it gives a basic salt Y with molecular formula XOH (Molecular mass = 40) and liberates a gas Z which easily catches fire. Identify X, Y and Z and also write the reaction involved.
Which of the following reacts with cold water vigorously?
The metal which produces hydrogen gas on reaction with dilute hydrochloric acid as well as sodium hydroxide solution is
Answer the questions based on the figure below:
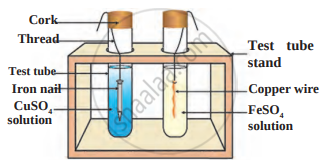
- Which experiment setup is demonstrated in the figure?
- What do you conclude after the reactions? Name reaction.
Keerti added dilute Hydrochloric acid to four metals and recorded her observations as shown in the table given below:
| Metal | Gas Evolved |
| Copper | Yes |
| Iron | Yes |
| Magnesium | No |
| Zinc | Yes |
Select the correct observation(s) and give chemical equation(s) of the reaction involved.
Select the appropriate state symbols of the products given as X and Y in the following chemical equation by choosing the correct option from table given below:
\[\ce{Zn_{(s)} + H_2SO_{4(l)} ->ZnSO_{4(X)} + H_{2(Y)}}\]
