(English Medium)
Academic Year: 2022-2023
Date & Time: 20th March 2023, 11:00 am
Duration: 2h
Advertisements
- Answers to this Paper must be written on the paper provided separately.
- You will not be allowed to write during the first 15 minutes.
- This time is to be spent reading the question paper.
- The time given at the head of this Paper is the time allowed for writing the answers.
- Attempt all questions from Section A and any four questions from Section B.
- The intended marks for questions or parts of questions are given in brackets [ ].
An element in period 3, whose electron affinity is zero:
Sulphur
Sulphur
Neon
Argon
Chapter: [0.01] Periodic Table, Periodic Properties and Variations of Properties
An element with the largest atomic radius among the following is ______.
Carbon
Nitrogen
Lithium
Beryllium
Chapter: [0.01] Periodic Table, Periodic Properties and Variations of Properties
The compound that is not an ore of aluminum is ______.
Cryolite
Corundum
Fluorspar
Bauxite
Chapter: [0.07] Metallurgy
The vapour density of CH3OH is ______. (At. Wt. C = 12, H = 1, O = 16)
32
18
16
34
Chapter: [0.05] Mole Concept and Stoichiometry
Which of the following reactions takes place at the anode during the electroplating of an article with silver?
\[\ce{Ag - 1e^{-} -> Ag^{1+}}\]
\[\ce{Ag + 1e^{-} -> Ag^{1-}}\]
\[\ce{Ag - 1e^{-} -> Ag}\]
None of the above
Chapter: [0.06] Electrolysis
The metallic hydroxide which forms a deep inky blue solution with excess ammonium hydroxide solution is ______.
Fe(OH)2
Cu(OH)2
Ca(OH)2
Fe(OH)3
Chapter: [0.08199999999999999] Ammonia
An example of a cyclic organic compound is ______.
Propene
Pentene
Butene
Benzene
Chapter: [0.09] Organic Chemistry
In the laboratory preparation, HCl gas is dried by passing through ______.
Dilute nitric acid
Concentrated sulphuric acid
Dilute sulphuric acid
Acidified water
Chapter: [0.081] Hydrogen Chloride [0.1] Practical Work
The nitrate, which undergoes one thermal decomposition, leaves behind a residue that is yellow when hot and white when cold.
Lead nitrate
Ammonium nitrate
Copper nitrate
Zinc nitrate
Chapter: [0.1] Practical Work
The salt is formed when concentrated sulphuric acid reacts with KNO3 above 200°C.
K2SO4
K2SO3
KHSO4
KHSO3
Chapter: [0.084] Sulphuric Acid
The property exhibited by concentrated sulphuric acid when it is used to prepare hydrogen chloride gas from potassium chloride is ______.
Dehydrating property
Drying property
Oxidising property
Non-volatile acid property
Chapter: [0.084] Sulphuric Acid
The hydrocarbon is formed when sodium propionate and soda lime are heated together.
Methane
Ethane
Ethene
Propane
Chapter: [0.09] Organic Chemistry
The acid which does not form acid salt by a basic radical.
H2CO3
H3PO4
H2SO4
CH3COOH
Chapter: [0.03] Study of Acids, Bases and Salts
The general formula of hydrocarbons with single covalent bonds is ______.
CnH2n+2
CnH2n
CnH2n–2
CnH2n–6
Chapter: [0.02] Chemical Bonding
The indicator that changes to pink colour in an alkaline solution is ______.
Blue Litmus
Methyl Orange
Red Litmus
Phenolphthalein
Chapter: [0.03] Study of Acids, Bases and Salts
Match the Column A with Column B:
| Column A | Column B |
| (a) Sodium Chloride | 1. has two shared pair of electrons |
| (b) Methane | 2. has high melting and boiling points |
| (c) Hydrogen chloride gas | 3. a greenhouse gas |
| (d) Oxidation reaction | 4. has low melting and boiling points |
| (e) Water | 5. \[\ce{Zn - 2e^- -> Zn^{2+}}\] |
| 6. \[\ce{S + 2e^{-} -> S^{2-}}\] |
Chapter:
The following sketch illustrates the process of conversion of Alumina to Aluminium:
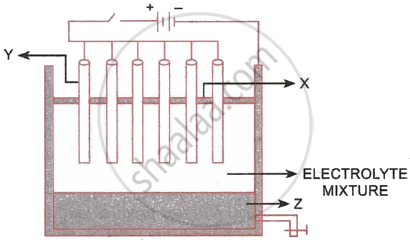
- Name the constituent of the electrolyte mixture which has a divalent metal in it.
- Name the powdered substances ‘X’ sprinkled on the surface of the electrolyte mixture.
- What is the name of the process?
- Write the reactions taking place at the electrodes ‘Y’ (anode) and ‘Z’ (cathode), respectively.
Chapter: [0.07] Metallurgy
Metals are good ______.
Oxidising agents
Reducing agents
Chapter: [0.06] Electrolysis
Non-polar covalent compounds are ______ conductors of heat and electricity.
Good
Bad
Chapter: [0.02] Chemical Bonding
Higher the pH value of a solution, the more ______ it is.
Acidic
Alkaline
Chapter: [0.03] Study of Acids, Bases and Salts [0.1] Practical Work
______ is a white precipitate that is soluble in excess of Ammonium hydroxide solution.
Silver chloride
Lead chloride
Chapter: [0.04] Analytical Chemistry
Conversion of ethene to ethane is an example of ______.
Hydration
Hydrogenation
Chapter: [0.09] Organic Chemistry
Give one word or phrase for the following:
The amount of energy released when an atom in the gaseous state accepts an electron to form an anion.
Chapter: [0.01] Periodic Table, Periodic Properties and Variations of Properties
Give one word/a phase/term/process for the following statement:
The tendency of an element to form chains of identical atoms.
Chapter: [0.09] Organic Chemistry
State the terms/process for the following:
The name of the process by which Ammonia is manufactured on a large scale.
Chapter: [0.08199999999999999] Ammonia
State the terms/process for the following:
A type of salt formed by partial replacement of hydroxyl radicals with an acid radical.
Chapter: [0.03] Study of Acids, Bases and Salts
Give one word or phrase for the following:
The ratio of the mass of a certain volume of gas to the mass of an equal volume of hydrogen under the same conditions of temperature and pressure.
Chapter: [0.05] Mole Concept and Stoichiometry
Give the structural formula of the following organic compound:
2-chlorobutane
Chapter: [0.09] Organic Chemistry
Give the structural formula of the following organic compound:
Methanal
Chapter: [0.09] Organic Chemistry
Give the structural formula of the following organic compound:
But-2-yne
Chapter: [0.09] Organic Chemistry
Give the IUPAC name of the following organic compound:
\[\begin{array}{cc}
\phantom{}\ce{H}\phantom{...}\ce{O}\phantom{.....}\\
\phantom{}|\phantom{....}||\phantom{.....}\\
\ce{H - C - C - O - H}\phantom{.}\\
|\phantom{..........}\\
\ce{H}\phantom{..........}
\end{array}\]
Chapter:
Give the IUPAC name of the following organic compound:
\[\begin{array}{cc}
\phantom{.}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{OH}\phantom{.}\ce{H}\phantom{.}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{}\\
\phantom{}\ce{H - C - C - C - C - H}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{}\\
\phantom{.}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{.}\\
\end{array}\]
Chapter:
Advertisements
Identify the cation in the following case:
Ammonium hydroxide solution when added to Solution B gives a white precipitate which does not dissolve in excess of ammonium hydroxide solution.
Chapter: [0.04] Analytical Chemistry
Identify the cation in the following case:
Sodium hydroxide solution when added to Solution C gives a white precipitate which is insoluble in excess of sodium hydroxide solution.
Chapter: [0.04] Analytical Chemistry
During electrolysis, the compound ______ in its molten state, liberates reddish brown fumes at the anode.
NaCl
PbBr2
Chapter: [0.06] Electrolysis
The ion which could be discharged most readily during electrolysis is ______.
Fe2+
Cu2+
Chapter: [0.06] Electrolysis
Arrange the following as per the instruction given in the bracket:
Al, K, Mg, Ca (decreasing order of its reactivity)
Chapter: [0.07] Metallurgy
Arrange the following as per the instruction given in the bracket:
N, Be, O, C (increasing order of non-metallic character)
Chapter:
Arrange the following as per the instruction given in the bracket:
P, Si, F, Be (decreasing order of valence electrons)
Chapter: [0.01] Periodic Table, Periodic Properties and Variations of Properties
Complete and balance the following equation:
\[\ce{NH4Cl + Ca(OH)2 ->}\]
Chapter: [0.05] Mole Concept and Stoichiometry
Complete and balance the following equation:
\[\ce{CuSO4 + NH3OH ->}\]
Chapter: [0.05] Mole Concept and Stoichiometry
Complete and balance the following equation:
\[\ce{Cu + Conc{.} HNO3 ->}\]
Chapter: [0.05] Mole Concept and Stoichiometry
State a relevant reason for the following:
Hydrogen chloride gas cannot be dried over quick lime.
Chapter: [0.081] Hydrogen Chloride [0.1] Practical Work
State a relevant reason for the following:
Ammonia gas is not collected over water.
Chapter: [0.08199999999999999] Ammonia
Name the alloys for the given composition:
Magnesium + Manganese + Aluminium + Copper
Chapter: [0.07] Metallurgy
Identify the alloy in the given composition.
iron, nickel, chromium, carbon
Chapter: [0.07] Metallurgy
Solve the following numerical problem:
Ethane burns in oxygen according to the chemical equation:
\[\ce{2C2H6 + 7O2 -> 4CO2 + 6H2O}\]
If 80 ml of ethane is burned in 300 ml of oxygen, find the composition of the resultant gaseous mixture when measured at room temperature.
Chapter: [0.05] Mole Concept and Stoichiometry
The following question pertains to the laboratory preparation of Ammonia gas from Magnesium nitride:
Write a balanced chemical equation for its preparation.
Chapter: [0.08199999999999999] Ammonia [0.1] Practical Work
The following question pertains to the laboratory preparation of Ammonia gas from Magnesium nitride:
Why is the method seldom used?
Chapter: [0.08199999999999999] Ammonia [0.1] Practical Work
The following question pertains to the laboratory preparation of Ammonia gas from Magnesium nitride:
How do you identify the gas that formed?
Chapter: [0.08199999999999999] Ammonia [0.1] Practical Work
Write one use of the following alloy:
Bronze
Chapter: [0.07] Metallurgy
Write one use of the following alloy:
Fuse metal
Chapter: [0.07] Metallurgy
Draw the electron dot structure for the following:
Ammonium ion
[At. No. : N = 7, H = 1]
Chapter: [0.02] Chemical Bonding
Draw the electron dot structure for the following:
A molecular nitrogen
[At. No. : N = 7, H = 1]
Chapter: [0.02] Chemical Bonding
Write balanced equations to show the preparation of the following:
Ethene from ethanol
Chapter: [0.09] Organic Chemistry
Write balanced equations to show the preparation of the following:
Ethyne from Calcium carbide
Chapter: [0.09] Organic Chemistry
Give a balanced chemical equation for the following conversion with condition:
Monochloromethane from methane
Chapter: [0.09] Organic Chemistry
Study the following observation and name the anion present in the reaction:
When a crystalline solid 'P' is warmed with concentrated H2SO4 and copper turnings a reddish brown gas is released.
Chapter: [0.1] Practical Work
Study the following observation and name the anion present in the reaction:
When few drops of dilute sulphuric acid is added to Salt 'R' and heated, a colourless gas is released which turns moist lead acetate paper silvery black.
Chapter: [0.1] Practical Work
Study the following observation and name the anion present in the reaction:
When few drops of barium nitrate solution is added to the salt solution 'Q', a white precipitate is formed which is insoluble in HCl.
Chapter: [0.1] Practical Work
Advertisements
Define the term Electronegativity.
Chapter: [0.01] Periodic Table, Periodic Properties and Variations of Properties
State Gay-Lussac's law of combining volumes.
Chapter: [0.05] Mole Concept and Stoichiometry
The Empirical formula of an organic compound is CHCl2.
If its relative molecular mass is 168, what is its molecular formula?
[At. Wt. C = 12, H = 1, Cl = 35.5]
Chapter: [0.05] Mole Concept and Stoichiometry
The metal that will not produce hydrogen gas when reacted with dilute acids.
Iron
Lead
Magnesium sulphite
Ferric chloride
Zinc
Copper
Sodium sulphide
Ferrous sulphate
Chapter: [0.03] Study of Acids, Bases and Salts
The compound that will produce sulphur dioxide gas when reacted with dilute HCl.
Iron
Lead
Magnesium sulphite
Ferric chloride
Zinc
Copper
Sodium sulphide
Ferrous sulphate
Chapter: [0.084] Sulphuric Acid
The solution of this compound produces dirty green precipitate with NaOH.
Iron
Lead
Magnesium sulphite
Ferric chloride
Zinc
Copper
Sodium sulphide
Ferrous sulphate
Chapter: [0.084] Sulphuric Acid
State one relevant observation for the following:
To the copper nitrate solution, initially, few drops of sodium hydroxide solution is added and then added in excess.
Chapter: [0.04] Analytical Chemistry
State one relevant observation for the following:
Burning of ammonia in excess of oxygen.
Chapter: [0.08199999999999999] Ammonia
State one relevant observation for the following:
Dry ammonia gas is passed over heated PbO.
Chapter: [0.1] Practical Work
Name the following:
Organic compounds with same molecular formula but different structural formula.
Chapter: [0.09] Organic Chemistry
Name the following:
Group of organic compounds where the successive members follow a regular structural pattern, successive compounds differ by a 'CH2' group.
Chapter: [0.09] Organic Chemistry
Give reason for the following:
Ionisation potential decreases down a group.
Chapter: [0.01] Periodic Table, Periodic Properties and Variations of Properties
Give reason for the following:
Ionic compounds do not conduct electricity in solid state.
Chapter:
Find the percentage of phosphorus in the fertilizer superphosphate Ca(H2PO4)2.
Chapter: [0.05] Mole Concept and Stoichiometry
Calculate:
Write the empirical formula of C8H18.
Chapter: [0.05] Mole Concept and Stoichiometry
Answer the following question with reference to the electrorefining of copper:
What is the anode made of?
Chapter: [0.07] Metallurgy
Answer the following question with reference to the electrorefining of copper:
What do you observe at the cathode?
Chapter: [0.07] Metallurgy
Answer the following questions with reference to the electrorefining of copper:
Write the reaction taking place at the cathode.
Chapter: [0.07] Metallurgy
Arrange the following according to the instruction given in bracket:
C2H2, C3H6, CH4, C2H4 (In the increasing order of the molecular weight)
Chapter: [0.05] Mole Concept and Stoichiometry
Arrange the following according to the instruction given in bracket:
Cu2+, Na+, Zn2+, Ag+ (The order of Preferential discharge at the cathode)
Chapter: [0.06] Electrolysis
Differentiate between the following pair based on the criteria given in the bracket:
Cane sugar and hydrated copper sulphate [using concentrated H2SO4]
Chapter: [0.084] Sulphuric Acid
Differentiate between the following pair based on the criteria given in the bracket:
Sulphuric acid and hydrochloric acid [type of salts formed]
Chapter: [0.081] Hydrogen Chloride
Convert the following reaction into a balanced chemical equation:
Ammonia to nitric oxide using oxygen and platinum catalyst.
Chapter: [0.083] Nitric Acid
Convert the following reaction into a balanced chemical equation:
Sodium hydroxide to sodium sulphate using sulphuric acid.
Chapter: [0.084] Sulphuric Acid
Convert the following reaction into a balanced chemical equation:
Ferrous sulphide to hydrogen sulphide using hydrochloric acid.
Chapter: [0.081] Hydrogen Chloride
A compound which undergoes thermal dissociation.
CCl4
PbO
NaCl
CuO
NH4Cl
Chapter: [0.08199999999999999] Ammonia
An amphoteric oxide.
CCl4
PbO
NaCl
CuO
NH4Cl
Chapter: [0.04] Analytical Chemistry
A compound which is a non-electrolyte.
CCl4
PbO
NaCl
CuO
NH4Cl
Chapter: [0.06] Electrolysis
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
CISCE previous year question papers ICSE Class 10 Chemistry with solutions 2022 - 2023
Previous year Question paper for CISCE ICSE Class 10 -2023 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Chemistry, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of CISCE ICSE Class 10 .
How CISCE ICSE Class 10 Question Paper solutions Help Students ?
• Question paper solutions for Chemistry will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.
