Advertisements
Advertisements
Question
Differentiate between the following pair based on the criteria given in the bracket:
Sulphuric acid and hydrochloric acid [type of salts formed]
Solution
| Sulphuric Acid | Hydrochloric Acid |
| Reacts with bases to form sulphate salts on neutralization. | Reacts with bases to form chloride salts on neutralization. |
APPEARS IN
RELATED QUESTIONS
State which of the two a solution of HCl in water or in toluene is an electrolyte. Explain.
State which of the two - a solution of HCl in water or in toluene is an electrolyte. Explain.
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
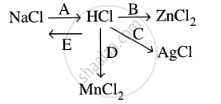
Convert Hydrochloric acid to nascent chlorine.
State which of the two — a solution of HCl in water or in toluene is an electrolyte. Explain.
State which of the two - a solution of HCl in water or in toluene is an electrolyte. Explain.
State which of the two - a solution of HCl in water or in toluene is an electrolyte. Explain.
Assertion (A): Dry hydrogen chloride gas is collected by the upward displacement of air.
Reason (R): Hydrogen chloride gas is lighter than air.
______ does not form an acid salt.
State which of the two - a solution of \[\ce{HCl}\] in water or in toluene is an electrolyte. Explain.
