Advertisements
Advertisements
Question
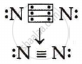
Draw the electron dot structure for the following:
A molecular nitrogen
[At. No. : N = 7, H = 1]
Solution
Nitrogen atoms share three electrons, making a triple bond.
APPEARS IN
RELATED QUESTIONS
Draw an electron dot diagram for the formation of the following. State the type of bonding present in them.
Ammonium ion
______ & ______ atoms have similar electronegativities.
Spot the error/correct the wrong statement:
Covalent compounds are soluble in polar solvents.
Covalent compounds have a low melting point.
Draw the electron dot structure of Methane molecule.
[Atomic number: N = 7, C = 6, H = 1]
Carbon tetrachloride is a ______ covalent molecule.
The following table shows the electronic configuration of the atoms A, B, C and D.
| Element | A | B | C | D |
| Electronic configuration | 2, 8, 8, 2 | 2, 6 | 2, 8, 7 | 2, 4 |
- Write the formula of the compound formed between:
- A and B
- D and C
- Which of the above elements will exhibit catenation?
Draw the structural formula of carbon tetrachloride and state the type of bond present in it.
Select the right answer.
Ammonium ion
Select the right answer.
Carbon tetrachloride
