Advertisements
Advertisements
Question
State a relevant reason for the following:
Hydrogen chloride gas cannot be dried over quick lime.
Solution
\[\ce{HCl}\] gas cannot be dried over quick lime because \[\ce{HCl}\] combines with \[\ce{CaO}\] to form salt and water.
\[\ce{2HCl + CaO -> CaCl2 + H2O}\]
APPEARS IN
RELATED QUESTIONS
The following are pertaining to the laboratory preparation of hydrogen chloride gas.
State a safety precaution you would take during the preparation of hydrochloric acid.
Name the acid used for the preparation of hydrogen chloride gas in the laboratory. Why is this particular acid preferred to other acids?
For the preparation of hydrochloric acid in the laboratory:
Why is the direct absorption of hydrogen chloride gas in water not feasible?
Give the balanced equation for the laboratory preparation of hydrogen chloride gas reaction.
Name the drying agents used in drying hydrogen chloride gas.
Explain why when the stopper of a bottle full of hydrogen chloride gas is opened, there are fumes in the air.
The given set up in the figure is for the preparation of an acid.
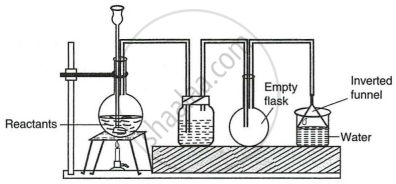
- Name the acid prepared by this method.
- Name the reactants used.
- Why an empty flask is used?
- What is the drying agent used? Why is this drying agent chosen?
- What is the role of the inverted funnel in the arrangement?
Draw a labelled diagram and explain the laboratory preparation of hydrogen chioride gas.
How will you prove that the gas prepared is HCI?
Explain, why (or give reasons for)
HCI gas does not conduct electricity, but hydrochloric acid conducts electricity.
What is aqua-regia?
Explain, why the following statement is not correct:
Lead chloride can be prepared by adding dilute HCI to lead sulphate solution.
Write the equation for the reaction which takes place in question(a).
Write the equation for :
The preparation of hydrogen chloride from sodium chloride and sulphuric acid. State whether the sulphuric acid should be concentrated or dilute.
Hydrogen chloride gas, being highly soluble in water, is dried by ______.
Give one test to distinguish between the following pair of chemicals.
Iron (III) Chloride solution and copper chloride solution.
Answer the following question related to the laboratory preparation of the hydrogen chloride gas:
State the temperature required in the preparation.
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl ->}\]
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl ->}\]
