Advertisements
Advertisements
प्रश्न
Assertion (A): Molar conductivity decreases with increase in concentration.
Reason (R): When concentration approaches zero, the molar conductivity is known as limiting molar conductivity.
पर्याय
Both (A) and (R) are true and (R) is the correct explanation of (A).
Both (A) and (R) are true, but (R) is not the correct explanation of (A).
(A) is true, but (R) is false.
(A) is false, but (R) is true.
उत्तर
Both (A) and (R) are true, but (R) is not the correct explanation of (A).
Explanation:
Molar conductivity increases as concentration lowers. This is because the total volume, V, of a solution containing one mole of electrolyte increases. A drop in 'κ' following the dilution of a solution has been discovered to be more than countered by an increase in volume. The limiting molar conductivity of a solution is its molar conductivity at infinite dilution. In other words, when the electrolyte concentration approaches zero, the molar conductivity is said to be limiting.
APPEARS IN
संबंधित प्रश्न
Define "Molar conductivity".
The molar conductivity of cation and anion of salt BA are 180 and 220 mhos respectively. The molar conductivity of salt BA at infinite dilution is_____________ .
(a) 90 mhos.cm2
(b) 110 mhos.cm2.mol-1
(c) 200 mhos.cm2.mol-1
(d) 400 mhos.cm2.mol-1
Why does the conductivity of a solution decrease with dilution?
Define the following terms :
Limiting molar conductivity
Consider figure and answer the question to given below.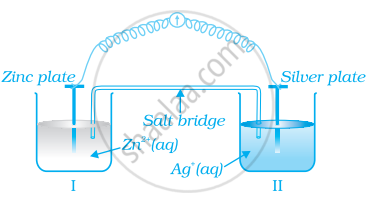
How will the concentration of Zn2+ ions and Ag+ ions be affected after the cell becomes ‘dead’?
An increase in equivalent conductance of a strong electrolyte with dilution is mainly due to :-
Which of the following halogen acids is the strongest reducing agent?
Given below are two statements:
Statements I: The limiting molar conductivity of KCl (strong electrolyte) is higher compared to that of CH3COOH (weak electrolyte).
Statement II: Molar conductivity decreases with decrease in concentration of electrolyte.
In the light of the above statements, choose the most appropriate answer from the options given below:
The following questions are case-based questions. Read the passage carefully and answer the questions that follow:
| Rahul set up an experiment to find the resistance of aqueous KCl solution for different concentrations at 298 K using a conductivity cell connected to a Wheatstone bridge. He fed the Wheatstone bridge with a.c. power in the audio frequency range 550 to 5000 cycles per second. Once the resistance was calculated from the null point, he also calculated the conductivity K and molar conductivity ∧m and recorded his readings in tabular form. |
| S. No. | Conc. (M) |
k S cm−1 | ∧m S cm2 mol−1 |
| 1. | 1.00 | 111.3 × 10−3 | 111.3 |
| 2. | 0.10 | 12.9 × 10−3 | 129.0 |
| 3. | 0.01 | 1.41 × 10−3 | 141.0 |
Answer the following questions:
(a) Why does conductivity decrease with dilution? (1)
(b) If `∧_"m"^0` of KCl is 150.0 S cm2 mol−1, calculate the degree of dissociation of 0.01 M KCI. (1)
(c) If Rahul had used HCl instead of KCl then would you expect the ∧m values to be more or less than those per KCl for a given concentration? Justify. (2)
OR
(c) Amit a classmate of Rahul repeated the same experiment with CH3COOH solution instead of KCl solution. Give one point that would be similar and one that would be different in his observations as compared to Rahul. (2)
The unit of molar conductivity is ______.
