Advertisements
Advertisements
प्रश्न
Balance the following equation. Also name the product formed.
` "H"_2"O"_2 → "H" _2 "O" + "O"_2`
उत्तर
`2 "H"_2 "O"_2 → 2"H"_2"O" +"O"_2` Water, oxygen
APPEARS IN
संबंधित प्रश्न
What is wrong with the following chemical equation?
Mg + O → MgO
Correct and balance it.
The metal M reacts vigorously with water to form a solution S and a gas G. The solution S turns red litmus to blue whereas gas G, which is lighter than air, burns with a pop sound. Metal M has a low melting point and it is used as a coolant in nuclear reactors.
(a) What is metal M?
(b) What is solution S? Is it acidic or alkaline?
(c) What is gas G?
(d) Write a balanced chemical equation for the reaction which takes place when metal M reacts with water.
(e) Is this reaction exothermic or endothermic?
Balance the following equation. Also name the product formed.
`"Zn" + "HCI" → "ZnCI" _2 + "H"_2`
Complete the following equation:
CH3COOH + NaOH →
Write the balanced chemical equation of the following reaction. iron + sulphuric acid → ferrous sulphate + hydrogen.
A chemical reaction is generally accompanied by certain external indications or characteristics. These include – change of – (a) colour (b) state (c) smell (d) evolution of gas (e) formation of precipitate (f) evolution or absorption of heat. With reference to change of colour – state the change in colour seen when the following are heated – copper carbonate.
Balance the following simple equation:
KHCO3 → K2CO3 + H2O + CO2
Write a balanced equation for the following word equation:
Potassium nitrate → Potassium nitrite + Oxygen
Complete the given chemical reaction.
\[\ce{CuSO_{4(aq)} + Fe_{(s)} ->}\]______ + ______
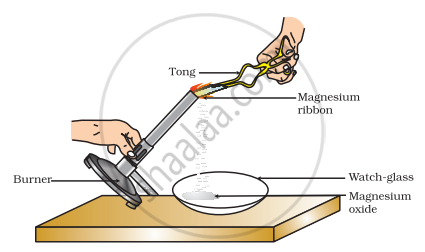
Which of the following is the correct observation of the reaction shown in the above set up?
