Advertisements
Advertisements
प्रश्न
Balance the following ionic equations.
\[\ce{Cr2O^{2-}7 + H^{+} + I- -> Cr^{3+} + I2 + H2O}\]
उत्तर
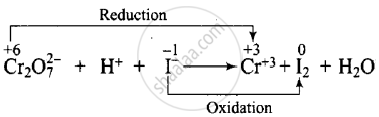
Dividing the equation into two half-reactions:
Oxidation half-reaction: \[\ce{I- -> I2}\]
Reduction half-reaction: \[\ce{CrO7^{2-} -> Cr^{3+}}\]
Balancing oxidation and reduction half-reactions separately as:
Oxidation half-reaction:
\[\ce{I- -> I2}\]
\[\ce{2I- -> I2}\]
\[\ce{2I- -> I2 + 2e-}\] ......(i)
Reduction half-reaction:
\[\ce{CrO^{2-}7 -> Cr^{3+}}\]
\[\ce{Cr2O^{2-}7 -> 2Cr^{3+}}\]
\[\ce{Cr2O^{2-}7 + 6e- -> 2Cr^{3+}}\]
\[\ce{Cr2O^{2-}7 + 14H^{+} + 6e- -> \underset{(acidic medium)}{2Cr^{3+} + 7H2O}}\] .....(ii)
To balance the electrons, multiply equation (i) by 3 and add to equation (ii)
\[\ce{Cr2O^{2-}7 + 14H^{+} + 6I- -> 2Cr^{3+} + 3I2 + 7H2O}\]
APPEARS IN
संबंधित प्रश्न
Calculate the oxidation number of sulphur, chromium and nitrogen in H2SO5, `"Cr"_2"O"_7^(2-)` and `"NO"_3^-`. Suggest structure of these compounds. Count for the fallacy.
Balance the following equation in the basic medium by ion-electron method and oxidation number methods and identify the oxidising agent and the reducing agent.
\[\ce{N2H4(l) + ClO^-_3 (aq) → NO(g) + Cl–(g)}\]
Chlorine is used to purify drinking water. Excess of chlorine is harmful. The excess of chlorine is removed by treating with sulphur dioxide. Present a balanced equation for this redox change taking place in water.
Balance the following redox equation by half-reaction method.
\[\ce{Bi(OH)_{3(s)} + SnO^2-_{2(aq)}->SnO^2-_{3(aq)} + Bi^_{(s)}(basic)}\]
Identify coefficients 'x' and 'y' for the following reaction.
\[\ce{{x}H2O2_{(aq)} + ClO^-_{4(aq)} -> 2O2_{(g)} + ClO^-_{2(aq)} + {y}H2O_{(l)}}\]
What is the change in oxidation number of Sulphur in following reaction?
\[\ce{MnO^-_{4(aq)} + SO^{2-}_{3(aq)} -> MnO^{2-}_{4(aq)} + SO^{2-}_{4(aq)}}\]
Write balanced chemical equation for the following reactions:
Reaction of liquid hydrazine \[\ce{(N2H4)}\] with chlorate ion \[\ce{(ClO^{-}3)}\] in basic medium produces nitric oxide gas and chloride ion in gaseous state.
Identify the redox reactions out of the following reactions and identify the oxidising and reducing agents in them.
\[\ce{HgCl2 (aq) + 2KI (aq) -> HgI2 (s) + 2KCl (aq)}\]
Identify the redox reactions out of the following reactions and identify the oxidising and reducing agents in them.
\[\ce{4NH3 (g) + 3O2 (g) -> 2N2 (g) + 6H2O (g)}\]
The weight of CO is required to form Re2(CO)10 will be ______ g, from 2.50 g of Re2O7 according to given reaction
\[\ce{Re2O7 + CO -> Re2(CO)10 + CO2}\]
Atomic weight of Re = 186.2; C = 12 and O = 16.
