Advertisements
Advertisements
प्रश्न
Calculate Standard cell potential for a cell having following reaction, \[\ce{2Al(s) + 3Ni^{2+} -> 2Al^{3+} + 3Ni(s)}\]
`("E"_"Ni"^circ = - 0.25 "V" and "E"_"Al"^circ = 1.66 "V")`
पर्याय
- 0.50 V
0.41 V
1.41 V
0.50 V
MCQ
उत्तर
1.41 V
Explanation:
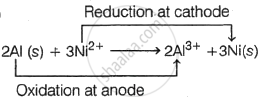
Given, `"E"_"Ni"^circ` = - 0.25 V
`"E"_"Al"^circ` = - 1.66 V
`"E"_"cell"^circ = "E"_"cathode"^circ - "E"_"anode"^circ`
= - 0.25 - (- 1.66)
= 1.41 V
shaalaa.com
Thermodynamics of Galvanic Cells
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
