Advertisements
Advertisements
प्रश्न
Calculate the number of unit cells in 0.3 g of a species having density of 8.5 g/cm3 and unit cell edge length 3.25 × 10-8 cm.
उत्तर
Given: Density (ρ) = 8.5 g/cm3, Edge length (a) = 3.25 × 10-8 cm, Mass of the species (x) = 0.3 g
To find: Number of unit cells in 0.3 g
Formula: Number of unit cells in x g of species = `x/(rho "a"^3)`
Calculation: Using formula,
Number of unit cells in 0.3 g of the species
`= (0.3 "g")/(8.5 "g cm"^-3 xx (3.25 xx 10^-8 "cm")^3)`
`= 1.03 xx 10^21`
Number of unit cells in 0.3 g of the given species is 1.03 × 1021.
संबंधित प्रश्न
Obtain the relationship between the density of a substance and the edge length of the unit cell.
An element with molar mass 27 g/mol forms a cubic unit cell with edge length of 405 pm. If the density of the element is 2.7 g/cm3, what is the nature of the cubic unit cell?
Give the percentage of empty space in bcc lattice.
If the total volume of a simple cubic unit cell is 6.817 × 10-23 cm3, what is the volume occupied by particles in the unit cell?
Derive the relationship between density of substance, its molar mass, and the unit cell edge length. Explain how you will calculate the number of particles, and a number of unit cells in x g of metal.
The pyranose structure of glucose is ____________.
What is the percentage of unoccupied space in fcc unit cell?
An element (atomic mass M g/mol) having bcc structure has unit cell edge 400 pm. The density of the element is ____________ g/cm3.
[NA = 6.0 × 1023 atom mol−1)
The number of atoms in 500 g of a fcc crystal of a metal with density d = 10 g/cm3 and cell edge 100 pm, is equal to ____________.
An element crystallizes bcc type of unit cell, the density and edge length of unit cell is 4 g cm−3 and 500 pm respectively. What is the atomic mass of an element?
Copper and silver have ____________ crystal structure.
Consider the following unit cell.
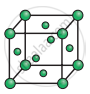
The number of particles (spheres) per unit cell is:
Which of the following formulae is used to find edge length of bee unit cell?
Gold crystallises into face-centred cubic cells. The edge length of a unit cell is 4.08 × 10–8 cm. Calculate the density of gold. [Molar mass of gold = 197 g mol–1]
An element has a bee structure with unit cell edge length of 288 pm. How many unit cells and number of atoms are present in 200 g of the element?
In face centred cubic unit cell, what is the volume occupied?
Calculate the density of metal with molar mass 56 g mol- 1 that crystallises to form a bcc structure with edge length 288 pm.
Silver crystallizes in the fcc structure. If the edge length of the unit cell is 400 pm, calculate the density of silver (Atomic mass of Ag = 108).
The correct sequence of the atomic layers in cubic close packing is ______.
The total number of different primitive unit cells is ______.
An element crystallises in fee structure. If molar mass of element is 72.7 g mol -1, the mass of its one unit cell will be ______.
