Advertisements
Advertisements
प्रश्न
Can `E_(Cell)^Θ` or `∆_rG^Θ` for cell reaction ever be equal to zero?
उत्तर
`E_(Cell)^Θ` or `∆_rG^Θ` can never be equal to zero. The only standard electrode potential which is arbitrarily assigned the value zero is the standard hydrogen electrode (SHE). Since everything else is measured concerning SHE; the EΘ cell can never be zero.
APPEARS IN
संबंधित प्रश्न
Calculate the potential of hydrogen electrode in contact with a solution whose pH is 10.
Calculate the emf of the cell in which the following reaction takes place:
\[\ce{Ni_{(s)} + 2Ag^+ (0.002 M) -> Ni^{2+} (0.160 M) + 2Ag_{(s)}}\]
Given that \[\ce{E^Θ_{cell}}\] = 1.05 V
Relationship between equilibrium constant of the reaction and standard electrode potential of electrochemical cell in which that reaction takes place is ____________.
Under what condition is ECell = 0 or ∆rG = 0?
Unlike dry cell, the mercury cell has a constant cell potential throughout its useful life. Why?
Assertion: Current stops flowing when ECell = 0.
Reason: Equilibrium of the cell reaction is attained.
Consider figure and answer the question to given below.
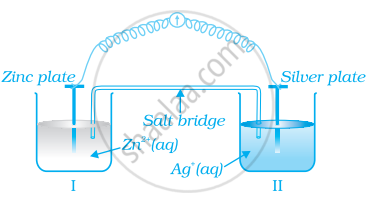
- Redraw the diagram to show the direction of electron flow.
- Is silver plate the anode or cathode?
- What will happen if salt bridge is removed?
- When will the cell stop functioning?
- How will concentration of Zn2+ ions and Ag+ ions be affected when the cell functions?
- How will the concentration of Zn2+ ions and Ag+ ions be affected after the cell becomes ‘dead’?
For the reaction H2 + 12 ⇌ 2Hl, the relation between equilibrium constants Kp and Kc is
For a cell reaction in involving a two electron change, the Stanford e. m. f of the cell is found to be 0.295 V at 25°C. The equilibrium constant of the reaction at 25°C will be
`"E"_"cell"^0` for the reaction, \[\ce{2H2O -> H3O+ + OH-}\] at 25°C is - 0.8277 V. The equilibrium constant for the reaction is ______.
