Advertisements
Advertisements
प्रश्न
Choose the correct answer:
The graph of PV vs P for gas is
पर्याय
Parabolic
Hyperbolic
A straight line parallel to the X-axis
A straight line passing through the origin
उत्तर
Straight-line parallel to the X-axis.
APPEARS IN
संबंधित प्रश्न
Three vessels of equal capacity have gases at the same temperature and pressure. The first vessel contains neon (monatomic), the second contains chlorine (diatomic), and the third contains uranium hexafluoride (polyatomic).
Is the root mean square speed of molecules the same in the three cases? If not, in which case is vrms the largest?
Name or state the following:
An equation used in chemical calculations which gives a simultaneous effect of changes of temperature and pressure on the volume of a given mass of dry gas
Give reason for the following:
Temperature remaining constant the product of the vol. & the press, of a given mass of dry gas is a constant.
Show that for monoatomic gas the ratio of the two specific heats is 5:3.
Gases exert pressure on the walls of the container because the gas molecules ______
Estimate the average thermal energy of a helium atom at the temperature on the surface of the Sun (6000 K).
Three vessels of equal capacity have gases at the same temperature and pressure. The first vessel contains neon (monatomic), the second contains chlorine (diatomic), and the third contains uranium hexafluoride (polyatomic).
Do the vessels contain an equal number of respective molecules?
Which of the following diagrams (Figure) depicts ideal gas behaviour?
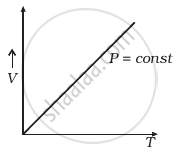 (a) |
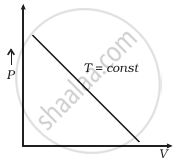 (b) |
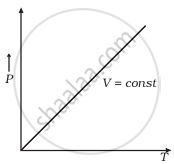 (c) |
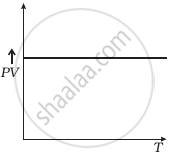 (d) |
Cooking gas containers are kept in a lorry moving with uniform speed. The temperature of the gas molecules inside will ______.
Two tanks of equal volume contain equal mass of oxygen and nitrogen at 127°C. Find the ratio of pressure in two tanks.
