Advertisements
Advertisements
प्रश्न
Choose the correct option from given alternative:
When a small amount of acid is added to water, the phenomena which occur are:
(A) Dilution
(B) Neutralization
(C) Formation of H3O+ ions
(D) Salt formation
पर्याय
(A) and (C)
(B) and (D)
(A) and (B)
(C) and (D)
उत्तर
The addition of acid to water is known as the process of dilution of acid, in this, the acid molecule loses H+ ions in the solution to form hydronium ion.
\[\ce{HCl +H2O -> H3O^+ + Cl^-}\]
Hence, the correct answer is an option (A) and (C).
APPEARS IN
संबंधित प्रश्न
While diluting an acid, why is it recommended that the acid should be added to water and not water to the acid?
What colour do the following indicators turn when added to a base or alkali (such as sodium hydroxide)?
red cabbage extract
Fill in the blank in the following sentences:
Acids produce.......................... ions on dissolving in water.
While diluting an acid, why is it recommended that the acid should be added to water and not water to the acid?
Which gas is liberated when dilute hydrochloric acid reacts with sodium carbonate?
10 mL of a solution of NaOH is found to be completely neutralised by 8 mL of a given solution of HCl. If we take 20 mL of the same solution of NaOH, the amount of HCl solution (the same solution as before) required to neutralise it will be:
(a) 4 mL
(b) 8 mL
(c) 12 mL
(d) 16 mL
What is common in all the water soluble bases (or alkalis)?
In the experimental set-up to show that "the germinating seeds give out carbon dioxide", answer the following questions:
(i) Why do we keep the conical flask airtight?
(ii) Name the substance kept in the small test tube inside the conical flask. Write its role.
(iii) Why does water rise in the delivery tube?
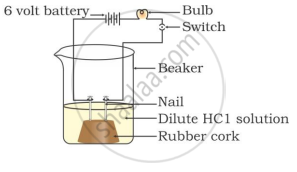
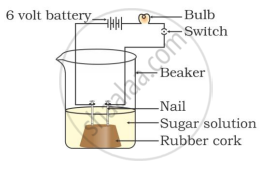
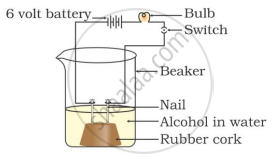
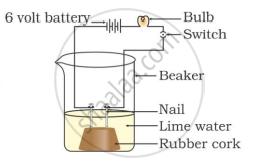
In which of the following setups would the bulb glow?
Which of the following is(are) true when HCl (g) is passed through water?
- It does not ionise in the solution as it is a covalent compound.
- It ionises in the solution
- It gives both hydrogen and hydroxyl ions in the solution
- It forms hydronium ion in the solution due to the combination of hydrogen ion with water molecule