Advertisements
Advertisements
प्रश्न
Choose the correct option from given alternative:
When a small amount of acid is added to water, the phenomena which occur are:
(A) Dilution
(B) Neutralization
(C) Formation of H3O+ ions
(D) Salt formation
विकल्प
(A) and (C)
(B) and (D)
(A) and (B)
(C) and (D)
उत्तर
The addition of acid to water is known as the process of dilution of acid, in this, the acid molecule loses H+ ions in the solution to form hydronium ion.
\[\ce{HCl +H2O -> H3O^+ + Cl^-}\]
Hence, the correct answer is an option (A) and (C).
APPEARS IN
संबंधित प्रश्न
What colour do the following indicators turn when added to a base or alkali (such as sodium hydroxide)?
(a) methyl orange
What happens when dilute hydrochloric acid is added to sodium carbonate? Write a balanced chemical equation of the reaction involved.
How will you test for the presence of this gas?
What is common in all the water soluble bases (or alkalis)?
What is meant by strong bases and weak bases? Classify the following into strong bases and weak bases:
NH4OH, Ca(OH)2, NaOH, KOH, Mg(OH)2
What happens when carbon dioxide gas is passed through lime water for a considerable time ?
Write equations of the reactions involved.
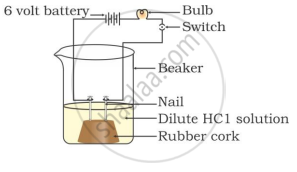
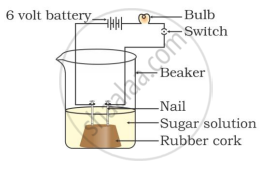
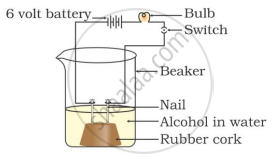
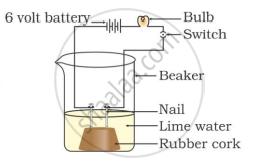
In which of the following setups would the bulb glow?
Which of the following phenomena occur when a small amount of acid is added to water?
- Ionisation
- Neutralisation
- Dilution
- Salt formation
Identify the correct representation of reaction occurring during chloralkali process
- A compound 'A' with a molecular formula of \[\ce{C2H4O2}\] reacts with a base to give salt and water. Identify 'A', state its nature and the name of the functional group it possesses. Write chemical equation for the reaction involved.
- When the above stated compound 'A' reacts with another compound 'B' having molecular formula \[\ce{C2H6O}\] in the presence of an acid, a sweet smelling compound is 'C' formed.
- Identify 'B' and 'C'.
- State the role of acid in this reaction.
- Write chemical equation for the reaction involved.