Advertisements
Advertisements
प्रश्न
How will you test for the presence of this gas?
उत्तर
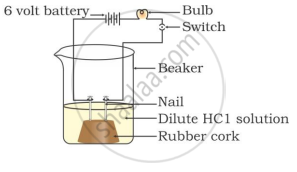
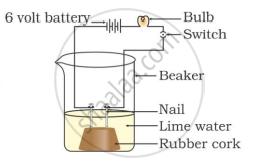
When the gas, liberated during the reaction, is passed through lime water, the lime water turns milky or a white precipitate of calcium carbonate is formed. This confirms that the gas is none other than carbon dioxide.
APPEARS IN
संबंधित प्रश्न
Why does distilled water not conduct electricity, whereas rain water does?
Why do acids not show acidic behaviour in the absence of water?
What colour do the following indicators turn when added to a base or alkali (such as sodium hydroxide)?
(a) methyl orange
What colours do the following indicators turn when added to an acid (such as hydrochloric acid)?
methyl orange
Fill in the blank in the following sentences:
Acids produce.......................... ions on dissolving in water.
You have been provided with three test-bubes. One of these test-tubes contains distilled water and the other two contain an acidic and a basic solution respectively. If you are given only blue litmus paper, how will you identify the contents of each test-tube?
What happens when carbon dioxide gas is passed through lime water for a considerable time ?
Write equations of the reactions involved.
In which of the following setups would the bulb glow?
Which of the following phenomena occur when a small amount of acid is added to water?
- Ionisation
- Neutralisation
- Dilution
- Salt formation
- A compound 'A' with a molecular formula of \[\ce{C2H4O2}\] reacts with a base to give salt and water. Identify 'A', state its nature and the name of the functional group it possesses. Write chemical equation for the reaction involved.
- When the above stated compound 'A' reacts with another compound 'B' having molecular formula \[\ce{C2H6O}\] in the presence of an acid, a sweet smelling compound is 'C' formed.
- Identify 'B' and 'C'.
- State the role of acid in this reaction.
- Write chemical equation for the reaction involved.