Advertisements
Advertisements
प्रश्न
Why do acids not show acidic behaviour in the absence of water?
उत्तर १
Acids do not show acidic behaviour in the absence of water because the dissociation of hydrogen ions from an acid occurs only in the presence of water. It is the hydrogen ions that are responsible for the acidic behaviour.
उत्तर २
Acids ionise only in the presence of water to give ions.
\[\ce{HCl + H2O -> H3O+ + Cl-}\]
However, in the absence of water, acids do not ionise to give H3O+ ions and therefore, do not behave as acids.
APPEARS IN
संबंधित प्रश्न
While diluting an acid, why is it recommended that the acid should be added to water and not water to the acid?
Compounds such as alcohols and glucose also contain hydrogen but are not categorized as acids. Describe an activity to prove it.
Why does distilled water not conduct electricity, whereas rain water does?
What colour do the following indicators turn when added to a base or alkali (such as sodium hydroxide)?
red cabbage extract
What colours do the following indicators turn when added to an acid (such as hydrochloric acid)?
methyl orange
Fill in the blank in the following sentences:
Acids produce.......................... ions on dissolving in water.
While diluting an acid, why is it recommended that the acid should be added to water and not water to the acid?
What happens when dilute hydrochloric acid is added to sodium carbonate? Write a balanced chemical equation of the reaction involved.
Which gas is liberated when dilute hydrochloric acid reacts with sodium carbonate?
How will you test for the presence of this gas?
What happens when carbon dioxide gas is passed through lime water for a short time?
Write equations of the reactions involved.
What is meant by strong bases and weak bases? Classify the following into strong bases and weak bases:
NH4OH, Ca(OH)2, NaOH, KOH, Mg(OH)2
What happens when carbon dioxide gas is passed through lime water for a considerable time ?
Write equations of the reactions involved.
Write the main difference between an acid and a base.
In the experimental set-up to show that "the germinating seeds give out carbon dioxide", answer the following questions:
(i) Why do we keep the conical flask airtight?
(ii) Name the substance kept in the small test tube inside the conical flask. Write its role.
(iii) Why does water rise in the delivery tube?
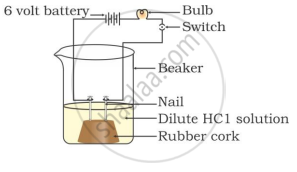
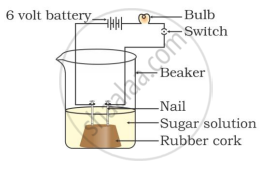
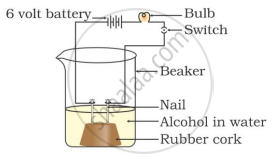
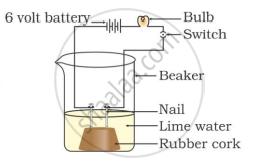
In which of the following setups would the bulb glow?
Which of the following phenomena occur when a small amount of acid is added to water?
- Ionisation
- Neutralisation
- Dilution
- Salt formation
Which of the following is(are) true when HCl (g) is passed through water?
- It does not ionise in the solution as it is a covalent compound.
- It ionises in the solution
- It gives both hydrogen and hydroxyl ions in the solution
- It forms hydronium ion in the solution due to the combination of hydrogen ion with water molecule
Identify the correct representation of reaction occurring during chloralkali process