Advertisements
Advertisements
प्रश्न
Why do acids not show acidic behaviour in the absence of water?
उत्तर १
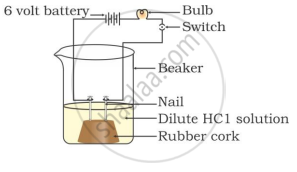
Acids do not show acidic behaviour in the absence of water because the dissociation of hydrogen ions from an acid occurs only in the presence of water. It is the hydrogen ions that are responsible for the acidic behaviour.
उत्तर २
Acids ionise only in the presence of water to give ions.
\[\ce{HCl + H2O -> H3O+ + Cl-}\]
However, in the absence of water, acids do not ionise to give H3O+ ions and therefore, do not behave as acids.
APPEARS IN
संबंधित प्रश्न
While diluting an acid, why is it recommended that the acid should be added to water and not water to the acid?
Why does distilled water not conduct electricity, whereas rain water does?
Lime water turns milky when __________ gas is passed through it.
(a) H2
(b) CO
(c) CO2
(d) SO2
What colour do the following indicators turn when added to a base or alkali (such as sodium hydroxide)?
(a) methyl orange
What colours do the following indicators turn when added to an acid (such as hydrochloric acid)?
methyl orange
Fill in the blank in the following sentences:
Acids produce.......................... ions on dissolving in water.
How will you test for the presence of this gas?
What happens when carbon dioxide gas is passed through lime water for a short time?
Write equations of the reactions involved.
You have been provided with three test-bubes. One of these test-tubes contains distilled water and the other two contain an acidic and a basic solution respectively. If you are given only blue litmus paper, how will you identify the contents of each test-tube?
Choose the correct option from given alternative:
When a small amount of acid is added to water, the phenomena which occur are:
(A) Dilution
(B) Neutralization
(C) Formation of H3O+ ions
(D) Salt formation
Answer the following question.
Blue litmus solution is added to two test tubes A and B containing dilute HCl and NaOH solution respectively. In which test tube a colour change will be observed? State the colour change and give its reason.
Vinay observed that the stain of curry on a white shirt becomes reddish-brown when soap is scrubbed on it, but it turns yellow again when the shirt is washed with plenty of water. What might be the reason for his observation?
- Soap is acidic in nature
- Soap is basic in nature
- Turmeric is a natural indicator which gives reddish tinge in bases
- Turmeric is a natural indicator which gives reddish tinge in acids
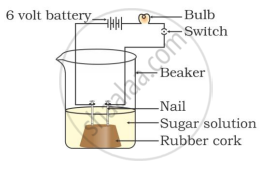
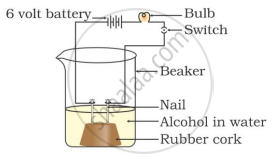
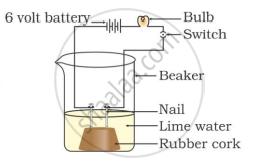
In which of the following setups would the bulb glow?
Which of the following phenomena occur when a small amount of acid is added to water?
- Ionisation
- Neutralisation
- Dilution
- Salt formation
Identify the correct representation of reaction occurring during chloralkali process
Salt A commonly used in bakery products on heating gets converted into another salt B which itself is used for the removal of hardness of water and a gas C is evolved. The gas C when passed through lime water, turns it milky. Identify A, B and C.
A metal carbonate X on reacting with an acid gives a gas which when passed through a solution Y gives the carbonate back. On the other hand, a gas G that is obtained at anode during electrolysis of brine is passed on dry Y, It gives a compound Z, used for disinfecting drinking water. Identity X, Y, G and Z.