Advertisements
Advertisements
प्रश्न
What happens when carbon dioxide gas is passed through lime water for a short time?
Write equations of the reactions involved.
उत्तर
Lime water turns milky.

APPEARS IN
संबंधित प्रश्न
Why do acids not show acidic behaviour in the absence of water?
What colours do the following indicators turn when added to an acid (such as hydrochloric acid)?
methyl orange
Explain with an example. Give the chemical equation of the reaction which takes place.
10 mL of a solution of NaOH is found to be completely neutralised by 8 mL of a given solution of HCl. If we take 20 mL of the same solution of NaOH, the amount of HCl solution (the same solution as before) required to neutralise it will be:
(a) 4 mL
(b) 8 mL
(c) 12 mL
(d) 16 mL
You have been provided with three test-bubes. One of these test-tubes contains distilled water and the other two contain an acidic and a basic solution respectively. If you are given only blue litmus paper, how will you identify the contents of each test-tube?
What is common in all the water soluble bases (or alkalis)?
What happens when carbon dioxide gas is passed through lime water for a considerable time ?
Write equations of the reactions involved.
Choose the correct option from given alternative:
When a small amount of acid is added to water, the phenomena which occur are:
(A) Dilution
(B) Neutralization
(C) Formation of H3O+ ions
(D) Salt formation
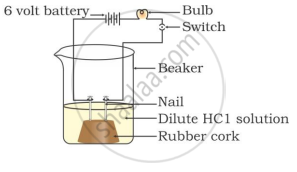
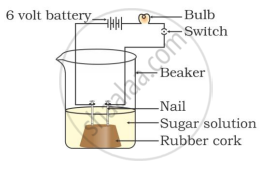
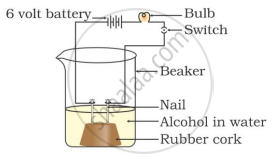
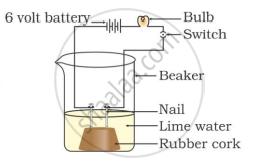
In which of the following setups would the bulb glow?
Which of the following phenomena occur when a small amount of acid is added to water?
- Ionisation
- Neutralisation
- Dilution
- Salt formation