Advertisements
Advertisements
प्रश्न
Compounds such as alcohols and glucose also contain hydrogen but are not categorized as acids. Describe an activity to prove it.
उत्तर
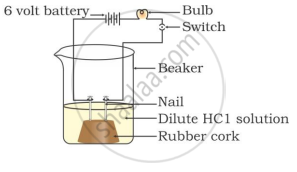
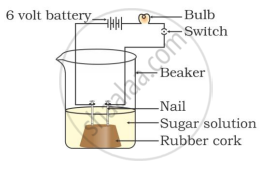
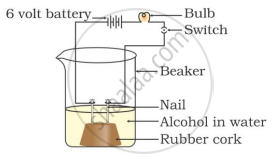
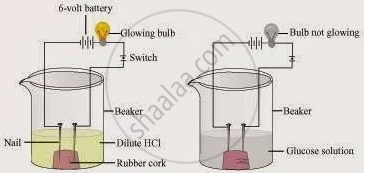
Two nails are fitted on a cork and kept in a 100-mL beaker. The nails are then connected to the two terminals of a 6-volt battery through a bulb and a switch. Some dilute HCl is poured into the beaker, and the current is switched on. The same experiment is then performed with a glucose solution and an alcohol solution.
Observations:
It will be observed that the bulb glows in the HCl solution and does not glow in the glucose solution.
Result:
HCl dissociates into H+ and Cl− ions. These ions conduct electricity in the solution, resulting in the glowing of the bulb. On the other hand, the glucose solution does not dissociate into ions. Therefore, it does not conduct electricity.
Conclusion:
From this activity, it can be concluded that all acids contain hydrogen, but not all compounds containing hydrogen are acids.
That is why, though alcohols and glucose contain hydrogen, they are not categorised as acids.
APPEARS IN
संबंधित प्रश्न
While diluting an acid, why is it recommended that the acid should be added to water and not water to the acid?
Why does distilled water not conduct electricity, whereas rain water does?
Why do acids not show acidic behaviour in the absence of water?
What colour do the following indicators turn when added to a base or alkali (such as sodium hydroxide)?
red cabbage extract
Fill in the blank in the following sentences:
Acids produce.......................... ions on dissolving in water.
While diluting an acid, why is it recommended that the acid should be added to water and not water to the acid?
Which gas is liberated when dilute hydrochloric acid reacts with sodium carbonate?
How will you test for the presence of this gas?
What is common in all the water soluble bases (or alkalis)?
What happens when carbon dioxide gas is passed through lime water for a considerable time ?
Write equations of the reactions involved.
Choose the correct option from given alternative:
When a small amount of acid is added to water, the phenomena which occur are:
(A) Dilution
(B) Neutralization
(C) Formation of H3O+ ions
(D) Salt formation
In which of the following setups would the bulb glow?
Which of the following phenomena occur when a small amount of acid is added to water?
- Ionisation
- Neutralisation
- Dilution
- Salt formation
Salt A commonly used in bakery products on heating gets converted into another salt B which itself is used for the removal of hardness of water and a gas C is evolved. The gas C when passed through lime water, turns it milky. Identify A, B and C.
A metal carbonate X on reacting with an acid gives a gas which when passed through a solution Y gives the carbonate back. On the other hand, a gas G that is obtained at anode during electrolysis of brine is passed on dry Y, It gives a compound Z, used for disinfecting drinking water. Identity X, Y, G and Z.
- A compound 'A' with a molecular formula of \[\ce{C2H4O2}\] reacts with a base to give salt and water. Identify 'A', state its nature and the name of the functional group it possesses. Write chemical equation for the reaction involved.
- When the above stated compound 'A' reacts with another compound 'B' having molecular formula \[\ce{C2H6O}\] in the presence of an acid, a sweet smelling compound is 'C' formed.
- Identify 'B' and 'C'.
- State the role of acid in this reaction.
- Write chemical equation for the reaction involved.