Advertisements
Advertisements
प्रश्न
Choose the correct option related to wavelengths (λ) of different parts of the electromagnetic spectrum.
पर्याय
`λ_"x-rays" < λ_"micro waves" < λ_"radio waves" < λ_"visible"`
`λ_"visible" < λ_"X-rays" < λ_"radio waves" < λ_"micro waves"`
`λ_"radio waves" < λ_"micro waves" < λ_"visible" <λ_"X-rays"`
`λ_"visible" < λ_"micro waves" < λ_"radio waves" < λ_"X-rays"`
उत्तर
`λ_"radio waves" < λ_"micro waves" < λ_"visible" <λ_"X-rays"`
Explanation:
∵ E = `(hc)/lambda`
The orderly arrangement of different parts of EM wave in decreasing order of wavelength is as follows:
`λ_"radio waves" > λ_"microwaves" > λ_"IR" > λ_"visible" > λ_"UV" > λ_"X-rays" > λ_"gamma"`
APPEARS IN
संबंधित प्रश्न
Given below are some famous numbers associated with electromagnetic radiations in different contexts in physics. State the part of the electromagnetic spectrum to which each belongs.
(a) 21 cm (wavelength emitted by atomic hydrogen in interstellar space).
(b) 1057 MHz (frequency of radiation arising from two close energy levels in hydrogen; known as Lamb shift).
(c) 2.7 K [temperature associated with the isotropic radiation filling all space-thought to be a relic of the ‘big-bang’ origin of the universe].
(d) 5890 Å - 5896 Å [double lines of sodium]
(e) 14.4 keV [energy of a particular transition in 57Fe nucleus associated with a famous high resolution spectroscopic method (Mössbauer spectroscopy)].
A wave has a wavelength of 10-3 nm. Name the wave.
Name the rays or waves of highest frequency .
An X-ray tube operates at 40 kV. Suppose the electron converts 70% of its energy into a photon at each collision. Find the lowest there wavelengths emitted from the tube. Neglect the energy imparted to the atom with which the electron collides.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
Name the part of the electromagnetic spectrum which is:
Produced by bombarding a metal target with high electrons.
What is the wavelength of the wave whose frequency is 1012 Hz? Name the electromagnetic wave.
Which of the following is a tool used for separating the different color wavelengths from each other?
Radio waves of constant amplitude can be generated with.
Identify the electromagnetic wave whose wavelength range is from about 10-3 m to about 10-1 m. Write one use of this.
In an atom X, electrons absorb the energy from an external source. This energy “excites” the electrons from a lower-energy level to a higher-energy level around the nucleus of the atom. When electrons return to the ground state, they emit photons.
The figure below is the energy level diagram of atom X with three energy levels, E1 = 0.00eV, E2 = 1.78eV and E3 = 2.95eV. The ground state is considered 0 eV for reference. The transition of electrons takes place between levels E1 and E2.
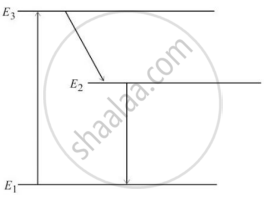
- What wavelength of radiation is needed to excite the atom to energy level E2 from E1?
- Suppose the external source has a power of 100 W. What would be the rate of photon emission?
