Advertisements
Advertisements
प्रश्न
Classify the substances given in Fig. 2.2 into elements and compounds
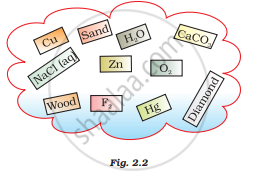
उत्तर
Elements – Cu, Zn, F2, O2, diamond (C), Hg
Compounds – CaCO3, H2O,
APPEARS IN
संबंधित प्रश्न
Fill in the blank
Chalk powder dissolved in water is an example of a Chalk powder dissolved in water is an example of a _________.
Match the columns.
| column A | column B |
|
1. to separate grain from chaff |
a. filtration |
| 2. to saparate sawdust from water | b. sedimentation and deeantation |
| 3. to separate iodine from sodium chloride | c. winnowing |
| 4. to separate iron fillings from sand | d.sublimation |
|
5. it is used to separate sand and water |
e maganetic separation |
|
|
f .handpicking |
Write the Chemical Name of the Following and Also Give Their Molecular Formulae:
Marble
Give reason: A frying pan is made up of steel but its handle is made up of wood.
Fill in the blank
A diatomic gaseous element is _______.
Give the name of the radical - SO4
Select the correct answer from the choice in the bracket.
The positively charged particle of an atom.
‘An atom is the basic unit of an element’. Draw a diagram of an atom – divisible as seen today.
‘The modern periodic table consists of elements arranged according to their increasing atomic numbers’. With reference to elements with atomic numbers 1 to 20 only in the periodic table – differentiate them into – metallic elements, metalloids, non-metals & noble gases.
Give an account of elements known to us.
