Advertisements
Advertisements
प्रश्न
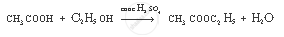
Complete the following equation:
`CH_3 COOH + CH_2 H_5 OH` 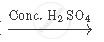
उत्तर
APPEARS IN
संबंधित प्रश्न
The molecular formula of acetic acid is _________ .
(a) CH3COOH
(b) CH3 – CH3
(c) C6H6
(d) C2H4
When you add a few drops of acetic acid to a test-tube containing sodium bicarbonate powder, which one of the following is your observation?
(A) No reaction takes place
(B) A colourless gas with pungent smell is released with brisk effervescence
(C) A brown coloured gas is released with brisk effervescence
(D) Formation of bubbles of a colourless and odourless gas
Name the gas evolved when ethanoic acid is added to sodium carbonate. How would you prove the presence of this gas?
How would you distinguish between ethanol and ethanoic acid by chemical test?
What do you notice when acetic acid reacts with metals?
Explain the following term with example.
Reduction
Explain the following term with an example.
Oxidant
Write the important uses of acetic acid.
The functional group present in acetic acid is ______.
Two statements are given - one labelled Assertion (A) and the other labelled Reason (R).
Assertion (A): Esterification is a process in which a sweet-smelling substance is produced.
Reason (R): When esters react with sodium hydroxide an alcohol and sodium salt of carboxylic acid are obtained.
