Advertisements
Advertisements
प्रश्न
Complete the following equation :
`2MnO_4^(-)+6H^++5NO_2^(-)rarr`
उत्तर
The complete chemical equation is given below.
`2MnO_4^(-)+6H^++5NO_2^(-)rarr 2Mn^(2+)+5NO_3^(-)+3H_2O`
APPEARS IN
संबंधित प्रश्न
Complete the following equations:
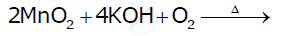
Describe the preparation of potassium dichromate from iron chromite ore. What is the effect of increasing pH on a solution of potassium dichromate?
Give an example and suggest a reason for the following feature of the transition metal chemistry:
A transition metal exhibits the highest oxidation state in oxides and fluorides.
Complete the following equations:
`2MnO_4^(-)+16H^++5S^(2-)rarr`
Using IUPAC norms write the formulae of Potassium trioxalatochromate (III)
When \[\ce{Cu^2+}\] ion is treated with \[\ce{KI}\], a white precipitate is formed. Explain the reaction with the help of chemical equation.
The product formed upon heating methyl bromide with potassium tert-butoxide is
Complete the reaction mentioning all the products formed:
\[\ce{Cr2O^{2-}7 + 3H2S + 8H^+ ->}\]
Indicate the steps in the preparation of \[\ce{K2Cr2O2}\] from chromite ore.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
