Advertisements
Advertisements
प्रश्न
When \[\ce{Cu^2+}\] ion is treated with \[\ce{KI}\], a white precipitate is formed. Explain the reaction with the help of chemical equation.
उत्तर
Reduction of \[\ce{Cu^2+}\] to \[\ce{Cu^+}\] takes place due to reaction with F ions.
This is given by the equation.
\[\ce{2Cu^2+ + 4I^- -> \underset{(White precipitate)}{Cu2I2 + I2}}\]
APPEARS IN
संबंधित प्रश्न
Complete the following equations:
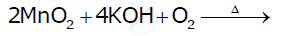
When chromite ore FeCr2O4 is fused with NaOH in presence of air, a yellow-coloured compound (A) is obtained, which on acidification with dilute sulphuric acid gives a compound (B). Compound (B) on reaction with KCl forms an orange coloured crystalline compound (C).
(i) Write the formulae of the compounds (A), (B) and C.
(ii) Write one use of compound (C).
Complete the following equation :
`2MnO_4^(-)+6H^++5NO_2^(-)rarr`
Write the formula of an oxo-anion of Manganese (Mn) in which it shows the oxidation state equal to its group number.
Complete the following chemical equations :
Cu + H2SO4(conc.) →
On addition of small amount of \[\ce{KMnO4}\] to concentrated \[\ce{H2SO4}\], a green oily compound is obtained which is highly explosive in nature. Identify the compound from the following.
Generally transition elements and their salts are coloured due to the presence of unpaired electrons in metal ions. Which of the following compounds are coloured?
(i) \[\ce{KMnO4}\]
(ii) \[\ce{Ce(SO4)2}\]
(iii) \[\ce{TiCl}\]
(iv) \[\ce{Cu2 Cl2}\]
When orange solution containing \[\ce{Cr2O^{2-}7}\] ion is treated with an alkali, a yellow solution is formed and when \[\ce{H^+}\] ions are added to yellow solution, an orange solution is obtained. Explain why does this happen?
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
Describe the preparation of potassium permanganate. How does the acidified permanganate solution react with oxalic acid? Write the ionic equation for the reaction.
