Advertisements
Advertisements
प्रश्न
Complete the statement by filling in the blank with the correct word:
Direct combination reaction of phosphorus pentoxide with water gives _______.
पर्याय
H3PO3
H3PO4
उत्तर
Direct combination reaction of phosphorus pentoxide with water gives H3PO4.
APPEARS IN
संबंधित प्रश्न
Find the odd man out:
Camphor, Ammonium Chloride, Naphthalene balls, Sugar
A silvery-white metal X taken in the form of ribbon, when ignited, burns in air with a dazzling white flame to form a white powder Y. When water is added to powder Y, it dissolves partially to form another substance Z.
(a) What could metal X be?
(b) What is powder Y?
(c) With which substance metal X combines to form powder Y?
(d) What is substance Z? Name one domestic use of substance Z.
(e) Write a balanced chemical equation of the reaction which takes place when metal X burns in air to form powder Y.
What type of reaction is represented by the following equation?
2 Ca + O2 → 2CaO
Consider the following equation of the chemical reaction of a metal M:
4M + 3O2 → 2M2O3
This equation represents:
(a) combination reaction as well as reduction reaction
(b) decomposition reaction as well as oxidation reaction
(c) oxidation reaction as well as displacement reaction
(d) combination reaction as well as oxidation reaction
Fill in the blank
A reaction in which two or more substances combine to form a single substance is called a ............ reaction.
(a) What is double displacement reaction? Explain with an example.
(b) A small amount of quick lime is added to water in a beaker.
(i) Name and define the type of reaction that has taken place.
(ii) Write balanced chemical equation for the above reaction and the chemical name of the product formed.
(iii) List two main observations of this reaction.
Select the correct answer for the statement given below:
The product formed during direct combination reaction of carbon dioxide and water.
The chemical reaction between copper and oxygen can be categorized as ______.
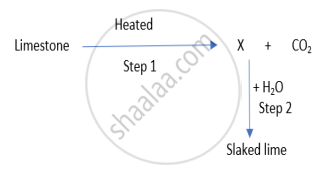
Identify the correct option from the given table which represents the type of reactions occurring in step 1 and step 2.
Identify the types of chemical reactions occurring during the combustion of fuel:
