Advertisements
Advertisements
प्रश्न
Consider the following equation of the chemical reaction of a metal M:
4M + 3O2 → 2M2O3
This equation represents:
(a) combination reaction as well as reduction reaction
(b) decomposition reaction as well as oxidation reaction
(c) oxidation reaction as well as displacement reaction
(d) combination reaction as well as oxidation reaction
उत्तर
(d) combination reaction as well as oxidation reaction
In the above reaction, metal combines with the oxygen to form a single compound, so it is a combination reaction. Moreover, addition of oxygen takes place, so it is also a oxidation reaction.
APPEARS IN
संबंधित प्रश्न
A solution of a substance ‘X’ is used for white washing.
- Name the substance ‘X’ and write its formula.
- Write the reaction of the substance ‘X’ named in (i) above with water.
what tpye of reaction is the following:
2K + Cl → 2KCl
When hydrogen burns in oxygen, water is formed and when water is electrolysed, then hydrogen and oxygen are produced. What type of reaction takes place:
(a) in the first case?
(b) in the second case
Explain the following type of chemical reaction, giving two examples for it:
Combination reaction
State why a direct combination reaction is called a – ‘synthesis reaction’.
Which changes occur during chemical changes?
The number of products formed in a synthesis reaction is ______
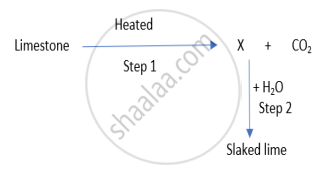
Identify the correct option from the given table which represents the type of reactions occurring in step 1 and step 2.
A metal ribbon 'X' bums in oxygen with a dazzling white flame forming a white ash 'Y'. The correct description of X, Y and the type of reaction is:
- Assertion: Rusting of Iron is endothermic in nature.
- Reason: As the reaction is slow, the release of heat is barely evident.
