Advertisements
Advertisements
प्रश्न
Explain the following type of chemical reaction, giving two examples for it:
Combination reaction
उत्तर
A combination reaction is a reaction in which two or more substances combine to form a single substance.
Examples:
- When iron and sulphur are heated together, they combine to form iron sulphide.
\[\ce{Fe + S ->[heat] FeS}\] - When carbon burns in oxygen, it forms a gaseous compound called carbon dioxide.
\[\ce{C + O2 ->[heat] CO2 + Heat}\]
APPEARS IN
संबंधित प्रश्न
What type of chemical reaction take place when a magnesium wire is burnt in air?
What do you understand by a chemical bond ?
Give one example each of which illustrates the following characteristics of a chemical reaction:
change of colour
What do you observe when solid lead nitrate is heated?
What effect does an increase in concentration of H+ (aq.) in a solution have on the pH of solution?
Define: Chemical combination reaction
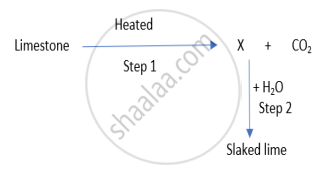
Identify the correct option from the given table which represents the type of reactions occurring in step 1 and step 2.
Identify the types of chemical reactions occurring during the combustion of fuel:
Read the text below and answer the questions that follow:
A small amount of hydrochloric acid was taken in a test tube. The test tube was heated. A glass rod was dipped in the ammonia solution and held on the top of the test tube. A white smoke was seen emanating from the tip of the glass rod.
- What must have happened?
- Which colour of gas is formed?
- Write the chemical equation for the reaction.
A clear solution of slaked lime is made by dissolving Ca(OH)2 in an excess of water. This solution is left exposed to air. The solution slowly goes milky as a faint white precipitate forms. Explain why a faint white precipitate forms, support your response with the help of a chemical equation.
