Advertisements
Advertisements
प्रश्न
A clear solution of slaked lime is made by dissolving Ca(OH)2 in an excess of water. This solution is left exposed to air. The solution slowly goes milky as a faint white precipitate forms. Explain why a faint white precipitate forms, support your response with the help of a chemical equation.
उत्तर
Calcium hydroxide reacts with Carbon dioxide present in the atmosphere to form Calcium carbonate which results in milkiness / white ppt / Formation of Calcium carbonate.
\[\ce{Ca(OH)2}\ + \ce{CO2}\ \ce{->}\ \ce{CaCO3}\ + \ce{H2O}\]
APPEARS IN
संबंधित प्रश्न
Define a combination reaction.
What happens during a chemical reaction ?
What do you understand by a chemical bond ?
What do you observe when solid lead nitrate is heated?
What do you observe when when dilute sulphuric acid is added to barium chloride solution ?
Fill in the blank
A reaction in which two or more substances combine to form a single substance is called a ............ reaction.
Classify the following reaction into different type:
CaO(s) + H2O(l) → Ca(OH)2(aq)
H2(g) + Cl29(g) → 2HCl(g) is a ______
The chemical reaction between copper and oxygen can be categorized as ______.
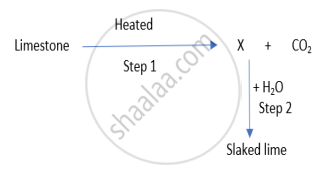
Identify the correct option from the given table which represents the type of reactions occurring in step 1 and step 2.
