Advertisements
Advertisements
प्रश्न
Name the functional group which always occurs in the middle of a carbon chain.
उत्तर
The functional group which always occurs in the middle of the carbon chain is the ketone group (-CO-). In a ketone, a carbon atom is bonded to one oxygen through a double bond. The two remaining free electrons in the carbon atom are shared with two different alkyl groups. This is why the -CO- group always occurs in the middle of the carbon chain.
APPEARS IN
संबंधित प्रश्न
Define the term ‘structural’ isomerism'.
List two reasons for carbon forming a large number of compounds. Name the type of bonding found in most of its compounds. Why does carbon form compounds mainly by this kind of bonding?
Draw the structures for Ethanoic acid.
Explain why propane cannot exhibit the structural isomerism property.
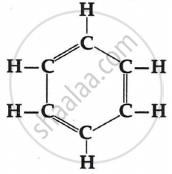
Which of the following is the molecular formula of benzene?
C6H6, C6H10, C6H12, C6H14
Which of the two has a branched chain : isobutane or normal butane?
Two organic compounds A and B have the same molecular formula C6H12. Write the names and structural formulae:
Which compound contains single bonds as well as a double bond?
Three organic compounds A, B and C have the following molecular formulae: C4H8O
Which molecular formula can represent an aldehyde as well as a ketone? Write the names and structural formulae of the aldehyde and ketone represented by this molecular formula.
Recognise the carbon chain type of the following:
\[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\\\ce{H - C - C - C - C - H}\\|\phantom{....}|\phantom{....}|\phantom{....}|\\
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}
\end{array}\]
Name the following: