Advertisements
Advertisements
प्रश्न
Define mass number of an element.
उत्तर
The total number of neutrons and protons in the nucleus is called the mass number of the element and is denoted by A.
APPEARS IN
संबंधित प्रश्न
What happens if the negative potential is changed on a grid?
State two similarities and two dissimilarities between the γ-rays and X-rays.
What happens to the mass number of an element when (i) an α -particle, (ii) a β -particle, and (iii) γ -radiation is emitted?
In the following fig. shows a radioactive source S in a thick lead container. The radiations pass through an electric field between the plates P and Q. Complete the diagram to show the paths of α , β and γ radiations.
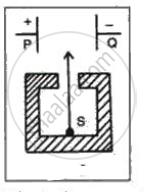
Why is the source S placed in a thick lead container?
A certain nucleus A (mass number 238 and atomic number 92) is radioactive and becomes a nucleus B (mass number 234 and atomic number 90) by the loss of one particle.
What particle was emitted?
During the emission of a beta particle, the ______ number remains same.
Justify with reason, whether the following nuclear reactions are allowed or not.
\[\ce{^A_Z X -> ^A_{Z - 2}X + ^4_{2}He}\]
State one use of radio-isotopes.
Mention one important function of ear main parts:
State the principle on which the functioning of a nuclear reactor is based.
The half-life of a radioactive nuclide is 100 hours. The fraction of original activity that will remain after 150 hours would be ______.
