Advertisements
Advertisements
प्रश्न
Define photoelectric effect.
उत्तर
The phenomenon of emission of electrons from a metal surface, when radiation of appropriate frequency is incident on it, is known as the photoelectric effect.
Explanation:
-
The emitted electrons are called photoelectrons.
-
The light must have a frequency greater than a threshold frequency (ν0) specific to the metal.
-
The energy of the incident light is used to overcome the work function (ϕ) of the metal and release electrons.
-
The kinetic energy of the emitted electrons is given by Einstein’s photoelectric equation:
hν = ϕ + Kmax
where:
- h = Planck’s constant
- ν = Frequency of incident light
- ϕ = Work function (minimum energy required to eject electrons)
- Kmax = Maximum kinetic energy of ejected electrons
The photoelectric effect provides evidence for the particle nature of light and led to the development of quantum mechanics.
संबंधित प्रश्न
Choose the correct option.
Polychromatic (containing many different frequencies) radiation is used in an experiment on the photoelectric effect. The stopping potential ______.
Explain the inverse linear dependence of stopping potential on the incident wavelength in a photoelectric effect experiment.
The electrons are emitted in the photoelectric effect from a metal surface.
What is the photoelectric effect? Define stopping potential and photoelectric work function.
The energy of a photon is 2 eV. Find its frequency and wavelength.
With the help of a circuit diagram describe the experiment to study the characteristics of the photoelectric effect. Hence discuss any 2 characteristics of the photoelectric effect.
The kinetic energy of the most energetic photoelectron emitted from a metal surface is doubled when the wavelength of the incident radiation is reduced from λ1 to λ2. The work function of the metal is ______
Which one of the following is TRUE in photoelectric emission?
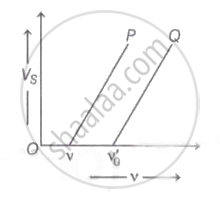
The graph of stopping potential `"V"_"s"` against frequency v of incident radiation is plotted for two different metals P and Q as shown in the graph. ΦP and ΦQ are work-functions of P and Q respectively, then
If the maximum kinetic energy of emitted electrons in photoelectric effect is 3.2 × 10-19 J and the work-function for metal is 6.63 × 10-19 J, then stopping potential and threshold wavelength respectively are
[Planck's constant, h = 6.63 × 1034 J-s]
[Velocity of light, c = 3 × 108 `"m"/"s"`]
[Charge on electron= 1.6 × 10-19 C]
Threshold frequency for a metal is 1015 Hz. Light of `lambda` = 4000 Å falls on its surface. Which of the following statements is correct?
In photoelectric effect, for a light of different intensities but of same frequency, the stopping potential for a given metal is ____________.
The photon of frequency vis incident on a metal surface whose threshold frequency is v0. The kinetic energy of the emitted photoelectrons will be ____________.
The lowest frequency of light that will cause the emission of photoelectrons from the surface of a metal (for which work function is 1.65 eV) will be ____________.
The work function of a photosensitive material is 4.0 eV. The longest wavelength of light that can cause photon emission from the substance is (approximately) ____________.
The radiations of energies 1 eV and 2.5 eV are incident on a metal surface having work function 0.5 eV. The ratio of the maximum velocities of the emitted photo-electrons is ____________.
A light of frequency 'v' is incident on the metal surface whose threshold frequency is 'v0'. If v = v0, then [c = speed of light in medium] ____________.
In experiment of photoelectric effect, the stopping potential for incident yellow light of wavelength 5890 Å is 4 volt. If the yellow light is replaced by blue light of wavelength 4000 Å, the stopping potential is ____________.
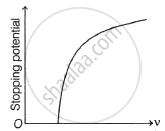
Which one of the following graphs represents the variation of photoelectric current (i) with intensity (I) of the incident light?
The stopping potential in the context of photoelectric effect depends on the following property of incident electromagnetic radiation ______.
A charged dust particle of radius 5 × 10-7 m is located in a horizontal electric field having an intensity of 6.28 × 105 V/m. The surrounding medium is air with a coefficient of viscosity η = 1.6 × 10-5 N-s/m2. If the particle moves with a uniform horizontal speed of 0.02 m/s, the number of electrons on it is ______.
Two radiations of photons energies 1 eV and 2.5 eV, successively illuminate a photosensitive metallic surface of work function 0.5 eV. The ratio of the maximum speeds of the emitted electrons is ______.
On a photosensitive material when frequency of incident radiation is increased by 30%, kinetic energy of emitted photoelectrons increases from 0.4 eV. The work function of the surface is ______.
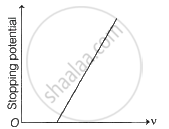
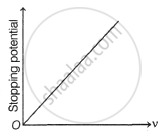
The following graphs show the variation of stopping potential corresponding to the frequency of incident radiation (ν) for a given metal. The correct variation is shown in graph [ν0 = threshold frequency].
|
(A) |
(B) |
|
(C) |
(D) |
Light of two different frequencies whose photons have energies 1.3 eV and 2.8 eV respectively, successfully illuminate a metallic surface whose work function is 0.8 eV. The ratio of maximum speeds of emitted electrons will be ______.
By increasing the voltage in an electron diffraction tube, the radius of the diffraction rings will ______.
The threshold frequency for a certain metal for photoelectric effect is 1.7 x 1015 Hz. When a light of frequency 2.2 x 1015 Hz is incident on the metal surface, the kinetic energy of the emitted photoelectrons is 3.3 x.10-19 J. Calculate Planck's constant.