Advertisements
Advertisements
प्रश्न
उत्तर
Take a round bottom flask which is filled with air and closed with a one holed rubber cork. A narrow capillary tube is passed through the cork and the flask is supported on a tripod in an inverted position as shown in the figure, so that the end of the capillary tube is underwater. Now, heat the flask gently with a spirit lamp. You will observe bubbles coming out from the capillary tube. This shows that gases expand on heating.

APPEARS IN
संबंधित प्रश्न
Give reason for Sea water does not freeze at 0°C.
How does the anomalous expansion of water help aquatic organisms in cold climates?
Calculate the time required to heat 20 kg of water from 10°C to 35°C using an immersion heater rated 1000 W. Assume that 80% of the power input is used to heat the water. Specific heat capacity of water = 42000 J kg−1 K−1.
Explain the following
Fishes survive in ponds even when the atmospheric temperature is well below 0°C.
In cold countries, the water pipes are covered with poor conductors. Why?
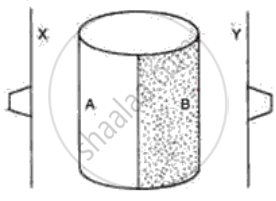
The following figure shows a metal cylinder, containing boiling water. One half side A is polished and another half, B is painted black. Two thin metal sheets X and Y are painted black and have one rubber stopper fixed with wax on each sheet. These sheets are equidistant from the boiling water (container A, B) as shown in the diagram. What would you expect to happen after a few minutes? Give a reason for your answer.
Observe the given graph and answer the following questions:
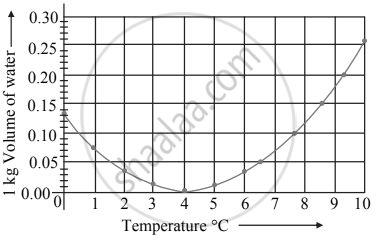
- Name the process represented in the figure.
- At what temperature does this process take place?
While studying anomalous behaviour of water in Hope’s apparatus, the upper temperature of the thermometer : 0 °C : : lower temperature of the thermometer : _______
Draw a neat and labelled diagram of Hope’s apparatus.
