Advertisements
Advertisements
प्रश्न
Discuss the main purpose of vulcanisation of rubber.
उत्तर १
Natural rubber though useful has some problems associated with its use. These limitations are discussed below:
1. Natural rubber is quite soft and sticky at room temperature. At elevated temperatures (> 335 K), it becomes even softer. At low temperatures (< 283 K), it becomes brittle. Thus, to maintain its elasticity, natural rubber is generally used in the temperature range of 283 K-335 K.
2. It has the capacity to absorb large amounts of water.
3. It has low tensile strength and low resistance to abrasion.
4. It is soluble in non-polar solvents.
5. It is easily attacked by oxidizing agents.
Vulcanization of natural rubber is done to improve upon all these properties. In this process, a mixture of raw rubber with sulphur and appropriate additive is heated at a temperature range between 373 K and 415 K.
उत्तर २
Natural rubber has the following disadvantages:
(a) It is soft and sticky and becomes even more so at high temperatures and brittle at low temperatures. Therefore, rubber is generally used in a narrow temperature range (283-335 K) where its elasticity is maintained.
(b)It has large water absorption capacity, has low tensile strength and low resistance to abrasion.
(c)It is not resistant to the action of organic solvents.
(d)It is easily attacked by oxygen and other oxidising agents. .
To improve all these properties, natural rubber is vulcanised by heating it with about 5% sulphur at 373-415 K. The vulcanized rubber thus obtained has excellent elasticity over a larger range of temperature, has low water absorption tendency and is resistant to the action of organic solvents and oxidising agents.
APPEARS IN
संबंधित प्रश्न
How does the presence of double bonds in rubber molecules influence their structure and reactivity?
Identify the monomer in the following polymeric structures.
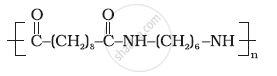
Write names and chemical formulae of monomers used in preparing Buna-N.
Write the structures of monomers used the following polymers:
Buna S
Complete the following statement by selecting the correct alternative from the choices given:
Natural rubber is a : _________
What is the role of Sulphur in the vulcanization of rubber?
Which of the following are example of synthetic rubber?
(i) Polychloroprene
(ii) Polyacrylonitrile
(iii) Buna-N
(iv) cis-polyisoprene
Vulcanisation makes rubber:
(i) more elastic
(ii) soluble in inorganic solvent
(iii) crystalline
(iv) more stiff
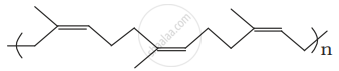
Identify the polymer given below:
Match materials given in Column I with the polymers given in Column II.
| Column I | Column II |
| (i) Natural rubber latex | (a) Nylon |
| (ii) Wood laminates | (b) Neoprene |
| (iii) Ropes and fibres | (c) Dacron |
| (iv) Polyester fabric | (d) Melamine formaldehyde resins |
| (v) Synthetic rubber | (e) Urea-formaldehyde resins |
| (vi) Unbreakable crockery | (f) cis-polyisoprene |
Which one of the following monomers gives the polymer neoprene on polymerization?
The polymer of natural rubber is:-
Which of the following statement is CORRECT regarding the drawbacks of raw rubber?
Match List I with List II.
| List I | List II |
| (Monomer Unit) | (Polymer) |
| (a) Caprolactum | (i) Natural rubber |
| (b) 2-Chloro-1, 3-butadiene | (ii) Buna-N |
| (c) Isoprene | (iii) Nylon-6 |
| (d) Acrylonitrile | (iv) Neoprene |
Choose the correct answer from the options given below:
