Advertisements
Advertisements
प्रश्न
Draw the structures of the following: H4P2O7 (Pyrophosphoric acid)
उत्तर
H4P2O7 (Pyrophosphoric acid)
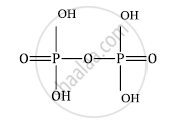
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
APPEARS IN
संबंधित प्रश्न
What is the basicity of H3PO4?
What is the basicity of H3PO3 ?
Complete the following equations : P4 + H2O →
What are the oxidation states of phosphorus in H3PO3
What are the oxidation states of phosphorus in the following:
PCl3
Phosphorus forms a number of oxoacids. Out of these oxoacids phosphinic acid has strong reducing property. Write its structure and also write a reaction showing its reducing behaviour.
Which among the oxoacids of phosphorus contain two P – OH bonds, one P = O and one P – H bond
What is the basicity of H3PO4?
What is the basicity of \[\ce{H3PO4}\]?
What is the basicity of H3PO4?
