Advertisements
Advertisements
प्रश्न
What is the basicity of H3PO3 ?
उत्तर
Basicity of H3PO3 is two as there are two replaceable hydrogen atoms. The structure of H3PO3 is as follows:
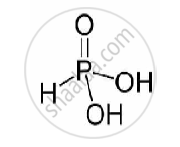
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
APPEARS IN
संबंधित प्रश्न
Give reasons for the following : H3PO2 is a stronger reducing agent than H3PO3.
What are the oxidation states of phosphorus in the following:
Ca3P2
What are the oxidation states of phosphorus in the following:
POF3
Write balanced chemical equations involved in the following reactions:
Calcium phosphide is dissolved in water.
Which of the following acids forms three series of salts?
Which one of the following is a dibasic acid?
What is the basicity of \[\ce{H3PO4}\]?
What is the basicity of \[\ce{H3PO4}\]?
What is the basicity of \[\ce{H3PO4}\]?
What is the basicity of \[\ce{H3PO4}\]?
