Advertisements
Advertisements
प्रश्न
What is the basicity of H3PO3 ?
उत्तर
Basicity of H3PO3 is two as there are two replaceable hydrogen atoms. The structure of H3PO3 is as follows:
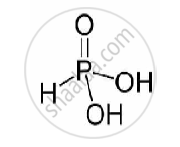
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
APPEARS IN
संबंधित प्रश्न
Draw the structures of the following: H4P2O7 (Pyrophosphoric acid)
What is the basicity of H3PO4?
Give the disproportionation reaction of H3PO3
What are the oxidation states of phosphorus in H3PO3
What are the oxidation states of phosphorus in the following:
PCl3
Which of the following acids forms three series of salts?
Which of the following contain P – O – P bond?
What is the basicity of H3PO4?
What is the basicity of \[\ce{H3PO4}\]?
What is the basicity of \[\ce{H3PO4}\]?
