Advertisements
Advertisements
प्रश्न
Each of the three states of matter has mass. Explain with the help simple experiments – that each state of matter has mass.
उत्तर
Matter i.e. solids, liquids, and Gases have mass explained by the following experiments.
(1) Solid
Experiment: A solid placed on one side of the scale, causes the scale to tilt towards one side.
Conclusion: The scale tilts due to the mass of the solid, hence all solids have mass.
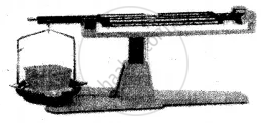
(2) Liquid:
Experiment: A liquid placed on one side of the scale, causes the scale to tilt towards one side.
Conclusion: The scale tilts due to the mass of the liquid, hence all liquids have mass.

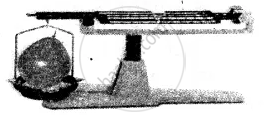
(3) Gas:
Experiment: An inflated balloon placed on one side of the scale causes it to tilt towards one side.
Conclusion: The scale tilts due to the mass of the gas, hence all gases have mass.
APPEARS IN
संबंधित प्रश्न
Define interconversion of states of matter. What are the two factors responsible for the change of states of matter?
Give reasons:
Liquids can flow
Give reason for the following.
Pure substances have fixed melting or boiling points.
Define the following term.
Element
Give two examples of the following:
Substances which do not change their states.
A kind of matter which can sublinje is
- water
- plastic
- milk
- iodine
Give reason of Why do liquids and gases flow but solids do not ?
Give reason of The odour of scent spreads in a room.
A piece of chalk can be broken into small particles by hammering but a piece of iron cannot be taken into small particles by hammering. Which characteristic of the particles of matter is illustrated by these observations?
Select the correct answer from A, B, C, D & E for the statement given below:
The state of matter, where the inter-particle attraction between particles is maximum.
