Advertisements
Advertisements
Question
Each of the three states of matter has mass. Explain with the help simple experiments – that each state of matter has mass.
Solution
Matter i.e. solids, liquids, and Gases have mass explained by the following experiments.
(1) Solid
Experiment: A solid placed on one side of the scale, causes the scale to tilt towards one side.
Conclusion: The scale tilts due to the mass of the solid, hence all solids have mass.
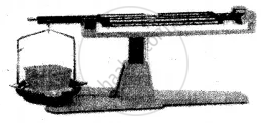
(2) Liquid:
Experiment: A liquid placed on one side of the scale, causes the scale to tilt towards one side.
Conclusion: The scale tilts due to the mass of the liquid, hence all liquids have mass.

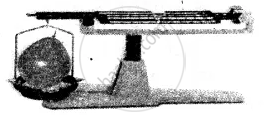
(3) Gas:
Experiment: An inflated balloon placed on one side of the scale causes it to tilt towards one side.
Conclusion: The scale tilts due to the mass of the gas, hence all gases have mass.
APPEARS IN
RELATED QUESTIONS
Give reasons for the following observation:
The smell of hot sizzling food reaches you several metres away, but to get the smell from cold food you have to go close.
Write your observation and conclusion for the following:
When one of the balloons suspended to the metre scale is punctured while other remains inflated ?
Write whether the following statement is true or false.
A gas has no free surface.
Write true or false for statement. Rewrite the false statement correctly.
Solids and liquids can flow.
What is the relation between intermolecular space and intermolecular force ?
Give reason Liquids can flow easily
Define the following term Intermolecular force .
Even two or three crystals of potassium permanganate can impart colour to a very large volume of water. Which characteristic of particles of matter is illustrated by this observation?
Match the characteristics of the three states of matter in List I with their correct answer from List II.
| List I | List II |
| 1. Have no definite shape, volume or free surface | A: Solids only |
| 2. Are highly compressible and least rigid | B: Liquids only |
| 3. Have a definite volume, no definite shape and are slightly diffusable | C: Gases only |
| 4. Are not compressible and have no diffusability | D: Liquids and gases only |
| 5. Have mass and occupy space | E: Solids and gases only |
| F: Solids, liquids and gases |
‘Inter-particle attraction between atoms of gases is very weak’. State five properties of gases which correlate as a consequence of the weak inter-particle attraction between particles of gases.
