Advertisements
Advertisements
प्रश्न
Equal lengths of magnesium ribbons are taken in test tubes A and B. Hydrochloric acid (HCl) is added to test tube A, while acetic acid (CH3COOH) is added to test tube B. In which test tube will the fizzing occur more vigorously, and why?
Equal lengths of magnesium ribbons are taken in test tubes A and B. Hydrochloric acid (HCl) is added to test tube A, while acetic acid (CH3COOH) is added to test tube B. The amount and concentration taken for both acids are the same. In which test tube will the fizzing occur more vigorously, and why?
उत्तर १
The fizzing will occur strongly in test tube A, in which hydrochloric acid (HCl) is added. This is because HCl is a stronger acid than CH3COOH and therefore produces hydrogen gas at a faster speed, due to which fizzing occurs.
उत्तर २
HCl is a strong acid, whereas acetic acid is a weaker acid. Fizzing occurs because the hydrogen gas evolves by reacting with the acid on the magnesium ribbon. Since HCl is a very strong acid, there is a lot of hydrogen gas liberated from test tube A. Therefore, more fizzing takes place in test tube A.
APPEARS IN
संबंधित प्रश्न
A solution reacts with crushed egg-shells to give a gas that turns lime-water milky. The solution contains ______.
Complete and balance the following chemical equations:
Zn (s) + HCI (aq) →
What happens when an acid reacts with a base?
What happens when an acid reacts with a metal oxide? Explain with the help of an example. Write a balanced equation for the reaction involved.
With the help of labelled diagrams, describe an activity to show that acids produce ions only in aqueous solutions.
Write a balanced chemical equation of the reaction which takes place.
A substance X which is used as an antacid reacts with dilute hydrochloric acid to produce a gas Y which is used in one type of fire-extinguisher. Name the substance X and gas Y. Write a balanced equation for the chemical reaction which takes place.
With which substance should chlorine be treated to get bleaching powder?
What happens when a solution of sodium hydrogencarbonate is heated? Write equation of the reaction involved.
When _______________ is passed through fresh lime water, it turns milky.
Acids turn blue litmus paper to ______.
Lime water reacts with chlorine to form:
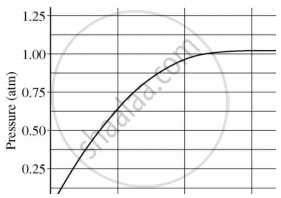
A student added 10 g of calcium carbonate in a rigid container, secured it tightly and started to heat it. After some time, an increase in pressure was observed, the pressure reading was then noted at intervals of 5 mins and plotted against time, in a graph as shown below. During which time interval did maximum decomposition take place?
What are strong and weak acids? In the following list of acids, separate strong acids from weak acids.
Hydrochloric acid, citric acid, acetic acid, nitric acid, formic acid, sulphuric acid.
A salt may be ______.
Solutions of acids conduct ______.
“All acids contain one or more hydrogen atoms, but not all substances containing hydrogen are acids.” Support this statement with examples.
______ & ______ metals do not react with HCl or HNO3.
What property do acids and bases have in common? Explain it with an example.
Consider the following salt:
\[\ce{ZCO3}\]
What would be the change in colour in blue litmus if \[\ce{ZCO3}\] is added to it and Z is potassium?
