Advertisements
Advertisements
प्रश्न
Complete and balance the following chemical equations:
Zn (s) + HCI (aq) →
उत्तर
Zn(s) + 2HCI(aq) → ZnCI2(aq)+H2(g)
APPEARS IN
संबंधित प्रश्न
On adding dilute hydrochloric acid to copper oxide powder, the solution formed is blue-green.
On the basis of the above reaction, what can you say about the nature of copper oxide?
A substance X which is used as an antacid reacts with dilute hydrochloric acid to produce a gas Y which is used in one type of fire-extinguisher. Name the substance X and gas Y. Write a balanced equation for the chemical reaction which takes place.
What ions are present in the solutions of following substances? (write the symbols only)
Sodium hydroxide
Complete and balance the following chemical equations:
`Ca(OH)_2 + Cl_2 ->`
Answer the following question.
Identify the acid and base which form sodium hydrogen carbonate. Write the chemical equation in support of your answer. State whether this compound is acidic, basic, or neutral. Also, write its pH value.
Acids turn blue litmus paper to ______.
Lime water reacts with chlorine to form:
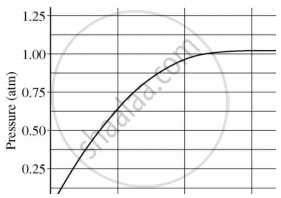
A student added 10 g of calcium carbonate in a rigid container, secured it tightly and started to heat it. After some time, an increase in pressure was observed, the pressure reading was then noted at intervals of 5 mins and plotted against time, in a graph as shown below. During which time interval did maximum decomposition take place?
A sulphate salt of Group 2 element of the Periodic Table is a white, soft substance which can be moulded into different shapes by making its dough. When this compound is left in open for some time, it becomes a solid mass and cannot be used for moulding purposes. Identify the sulphate salt. Why does it show such a behaviour? Give the reaction involved.
Write notes on the properties of acids.
