Advertisements
Advertisements
प्रश्न
Answer the following question.
Identify the acid and base which form sodium hydrogen carbonate. Write the chemical equation in support of your answer. State whether this compound is acidic, basic, or neutral. Also, write its pH value.
उत्तर
Sodium hydroxide(base) and hydrogen carbonate(acid) combine to form sodium hydrogen carbonate.
\[\ce{2NaOH + H2CO3 -> Na2CO3 + 2H2O}\]
Sodium hydrogen carbonate is a basic salt because NaOH is a strong base and H2CO3 is a weak acid.
pH value of sodium hydrogen carbonate is 8.27.
APPEARS IN
संबंधित प्रश्न
| Column A | Column B | ||
| i | eosin | 1 | losing hydrogen |
| ii | oxidation | 2 | synthetic indicator |
| 3 | losing oxygen | ||
| 4 | natural indicator |
When phenolphthalein is added to NaOH, the colour of the solution will become _________.
- colourless
- red
- pink
- yellow
Complete and balance the following chemical equations:
Zn (s) + HCI (aq) →
What happens when an acid reacts with a metal oxide? Explain with the help of an example. Write a balanced equation for the reaction involved.
What does a soda-acid type fire extinguisher contain? How does it work? Explain the working of a soda-acid fire extinguisher with the help of a labelled diagram.
Write any four physical properties of acids.
In an attempt to demonstrate electrical conductivity through an electrolyte, the apparatus setup. Which among the following statement(s) is(are) correct?
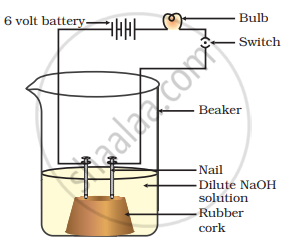
- Bulb will not glow because electrolyte is not acidic
- Bulb will glow because NaOH is a strong base and furnishes ions for conduction.
- Bulb will not glow because circuit is incomplete
- Bulb will not glow because it depends upon the type of electrolytic solution
Which of the following is used for dissolution of gold?
In the following schematic diagram for the preparation of hydrogen gas as shown in the figure, what would happen if following changes are made?

- In place of zinc granules, same amount of zinc dust is taken in the test tube
- Instead of dilute sulphuric acid, dilute hydrochloric acid is taken
- In place of zinc, copper turnings are taken
- Sodium hydroxide is taken in place of dilute sulphuric acid and the tube is heated.
Take a clean test tube with a holder and pour some dilute hydrochloric acid. Add a few pieces of magnesium ribbon pieces slowly. What do you observe? Now show a burning match stick near the mouth of the test tube. Do you hear any sound? The gas burns with a pop sound. From this, it is observed that hydrogen gas has been formed due to the reaction between acid and metal.
